Flipping lipids: why an' what's the reason for?
- PMID: 19689162
- PMCID: PMC2784161
- DOI: 10.1021/cb900163d
Flipping lipids: why an' what's the reason for?
Abstract
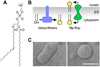
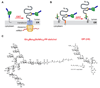
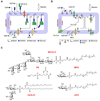
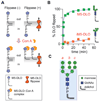
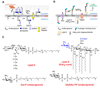
The biosynthesis of glycoconjugates such as N-glycoproteins and GPI-anchored proteins in eukaryotes and cell wall peptidoglycan and lipopolysaccharide in bacteria requires lipid intermediates to be flipped rapidly across the endoplasmic reticulum or bacterial cytoplasmic membrane (so-called biogenic membranes). Rapid flipping is also required to normalize the number of glycerophospholipids in the two leaflets of the bilayer as the membrane expands in a growing cell. Although lipids diffuse rapidly in the plane of the membrane, the intrinsic rate at which they flip across membranes is very low. Biogenic membranes possess dedicated lipid transporters or flippases to increase flipping to a physiologically sufficient rate. The flippases are "ATP-independent" and facilitate "downhill" transport. Most predicted biogenic membrane flippases have not been identified at the molecular level, and the few flippases that have been identified by genetic approaches have not been biochemically validated. Here we summarize recent progress on this fundamental topic and speculate on the mechanism(s) by which biogenic membrane flippases facilitate transbilayer lipid movement.
Figures

References
-
- Palade GE. Membrane biogenesis: an overview. Methods Enzymol. 1983;96:xxix–lv. - PubMed
-
- Blobel G. Protein targeting (Nobel lecture) Chembiochem. 2000;1:86–102. - PubMed
-
- White SH, von Heijne G. How translocons select transmembrane helices. Annu Rev Biophys. 2008;37:23–42. - PubMed
-
- Kornberg RD, McConnell HM. Inside-outside transitions of phospholipids in vesicle membranes. Biochemistry. 1971;10:1111–1120. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

