Changes in expression of sensory organ-specific microRNAs in rat dorsal root ganglia in association with mechanical hypersensitivity induced by spinal nerve ligation
- PMID: 19699278
- PMCID: PMC2762008
- DOI: 10.1016/j.neuroscience.2009.08.033
Changes in expression of sensory organ-specific microRNAs in rat dorsal root ganglia in association with mechanical hypersensitivity induced by spinal nerve ligation
Abstract
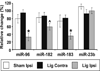
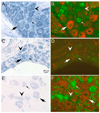
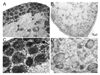
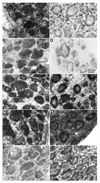
Chronic neuropathic pain caused by peripheral nerve injury is associated with global changes in gene expression in damaged neurons. To understand the molecular mechanisms underlying neuropathic pain, it is essential to elucidate how nerve injury alters gene expression and how the change contributes to the development and maintenance of chronic pain. MicroRNAs are non-protein-coding RNA molecules that regulate gene expression in a wide variety of biological processes mainly at the level of translation. This study investigated the possible involvement of microRNAs in gene regulation relevant to neuropathic pain. The analyses focused on a sensory organ-specific cluster of microRNAs that includes miR-96, -182, and -183. Quantitative real-time polymerase chain reaction (qPCR) analyses confirmed that these microRNAs were highly enriched in the dorsal root ganglion (DRG) of adult rats. Using the L5 spinal nerve ligation (SNL) model of chronic neuropathic pain, we observed a significant reduction in expression of these microRNAs in injured DRG neurons compared to controls. In situ hybridization and immunohistochemical analyses revealed that these microRNAs are expressed in both myelinated (N52 positive) and unmyelinated (IB4 positive) primary afferent neurons. They also revealed that the intracellular distributions of the microRNAs in DRG neurons were dramatically altered in animals with mechanical hypersensitivity. Whereas microRNAs were uniformly distributed within the DRG soma of non-allodynic animals, they were preferentially localized to the periphery of neurons in allodynic animals. The redistribution of microRNAs was associated with changes in the distribution of the stress granule (SG) protein, T-cell intracellular antigen 1 (TIA-1). These data demonstrate that SNL induces changes in expression levels and patterns of miR-96, -182, and -183, implying their possible contribution to chronic neuropathic pain through translational regulation of pain-relevant genes. Moreover, SGs were suggested to be assembled and associated with microRNAs after SNL, which may play a role in modification of microRNA-mediated gene regulation in DRG neurons.
Figures



Similar articles
-
Upregulation of interleukin-6 on Cav3.2 T-type calcium channels in dorsal root ganglion neurons contributes to neuropathic pain in rats with spinal nerve ligation.Exp Neurol. 2019 Jul;317:226-243. doi: 10.1016/j.expneurol.2019.03.005. Epub 2019 Mar 11. Exp Neurol. 2019. PMID: 30872136
-
Involvement of hyperpolarization-activated, cyclic nucleotide-gated cation channels in dorsal root ganglion in neuropathic pain.Sheng Li Xue Bao. 2008 Oct 25;60(5):579-80. Sheng Li Xue Bao. 2008. PMID: 18958363
-
Role of mitogen-activated protein kinase activation in injured and intact primary afferent neurons for mechanical and heat hypersensitivity after spinal nerve ligation.J Neurosci. 2004 Nov 10;24(45):10211-22. doi: 10.1523/JNEUROSCI.3388-04.2004. J Neurosci. 2004. PMID: 15537893 Free PMC article.
-
Spinal nerve ligation decreases γ-aminobutyric acidB receptors on specific populations of immunohistochemically identified neurons in L5 dorsal root ganglion of the rat.J Comp Neurol. 2012 Jun 1;520(8):1663-77. doi: 10.1002/cne.23005. J Comp Neurol. 2012. PMID: 22120979 Free PMC article.
-
The role of microRNAs in neurons and neuroimmune communication in the dorsal root ganglia in chronic pain.Neurosci Lett. 2020 Sep 14;735:135230. doi: 10.1016/j.neulet.2020.135230. Epub 2020 Jul 1. Neurosci Lett. 2020. PMID: 32621949 Review.
Cited by
-
Neuronal Differentiation of Human Mesenchymal Stem Cells Using Exosomes Derived from Differentiating Neuronal Cells.PLoS One. 2015 Aug 6;10(8):e0135111. doi: 10.1371/journal.pone.0135111. eCollection 2015. PLoS One. 2015. PMID: 26248331 Free PMC article.
-
Whiplash-Associated Dysphagia: Considerations of Potential Incidence and Mechanisms.Dysphagia. 2020 Jun;35(3):403-413. doi: 10.1007/s00455-019-10039-4. Epub 2019 Aug 3. Dysphagia. 2020. PMID: 31377863 Free PMC article.
-
MicroRNAs are potential prognostic and therapeutic targets in diabetic osteoarthritis.J Bone Miner Metab. 2015 Jan;33(1):1-8. doi: 10.1007/s00774-014-0628-0. Epub 2014 Sep 23. J Bone Miner Metab. 2015. PMID: 25245120 Review.
-
Computational functional genomics based analysis of pain-relevant micro-RNAs.Hum Genet. 2015 Nov;134(11-12):1221-38. doi: 10.1007/s00439-015-1600-3. Epub 2015 Sep 18. Hum Genet. 2015. PMID: 26385553
-
MicroRNA-146a is linked to pain-related pathophysiology of osteoarthritis.Gene. 2011 Jul 1;480(1-2):34-41. doi: 10.1016/j.gene.2011.03.003. Epub 2011 Mar 21. Gene. 2011. PMID: 21397669 Free PMC article.
References
-
- Abramoff MD, Magelhaes PJ, Ram SJ. Image Processing with ImageJ. Biophotonics International. 2004;11:36–42.
-
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. - PubMed
-
- Bhattacharyya SN, Habermacher R, Martine U, Closs EI, Filipowicz W. Relief of microRNA-mediated translational repression in human cells subjected to stress. Cell. 2006;125:1111–1124. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

