Design, synthesis, and biological evaluation of hydroquinone derivatives as novel inhibitors of the sarco/endoplasmic reticulum calcium ATPase
- PMID: 19699645
- PMCID: PMC2752035
- DOI: 10.1016/j.bmc.2009.07.075
Design, synthesis, and biological evaluation of hydroquinone derivatives as novel inhibitors of the sarco/endoplasmic reticulum calcium ATPase
Abstract
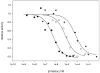
Analogues of the compound 2,5-di-tert-butylhydroquinone (BHQ) are capable of inhibiting the enzyme sarco/endoplasmic reticulum ATPase (SERCA) in the low micromolar and submicromolar concentration ranges. Not only are SERCA inhibitors valuable research tools, but they also have potential medicinal value as agents against prostate cancer. This study describes the synthesis of 13 compounds representing several classes of BHQ analogues, such as hydroquinones with a single aromatic substituent, symmetrically and unsymmetrically disubstituted hydroquinones, and hydroquinones with omega-amino acid tethers attached to their hydroxyl groups. Structure-activity relationships were established by measuring the inhibitory potencies of all synthesized compounds in bioassays. The assays were complemented by computational ligand docking for an analysis of the relevant ligand/receptor interactions.
Figures

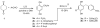
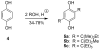
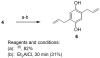
Similar articles
-
Molecular determinants of sarco/endoplasmic reticulum calcium ATPase inhibition by hydroquinone-based compounds.Proteins. 2008 Feb 15;70(3):639-49. doi: 10.1002/prot.21542. Proteins. 2008. PMID: 17879345
-
Structure-based virtual screening for novel inhibitors of the sarco/endoplasmic reticulum calcium ATPase and their experimental evaluation.Bioorg Med Chem. 2009 Feb 1;17(3):1353-60. doi: 10.1016/j.bmc.2008.12.010. Epub 2008 Dec 14. Bioorg Med Chem. 2009. PMID: 19117760
-
Comparison of current docking tools for the simulation of inhibitor binding by the transmembrane domain of the sarco/endoplasmic reticulum calcium ATPase.Biophys Chem. 2010 Aug;150(1-3):88-97. doi: 10.1016/j.bpc.2010.01.011. Epub 2010 Feb 4. Biophys Chem. 2010. PMID: 20167416 Free PMC article.
-
Long-range intramolecular linked functions in activation and inhibition of SERCA ATPases.Ann N Y Acad Sci. 1992 Nov 30;671:32-47; discussion 48. doi: 10.1111/j.1749-6632.1992.tb43782.x. Ann N Y Acad Sci. 1992. PMID: 1288328 Review. No abstract available.
-
Endoplasmic reticulum calcium transport ATPase expression during differentiation of colon cancer and leukaemia cells.Biochem Biophys Res Commun. 2004 Oct 1;322(4):1223-36. doi: 10.1016/j.bbrc.2004.08.030. Biochem Biophys Res Commun. 2004. PMID: 15336970 Review.
Cited by
-
Targeting oncogenic Notch signaling with SERCA inhibitors.J Hematol Oncol. 2021 Jan 6;14(1):8. doi: 10.1186/s13045-020-01015-9. J Hematol Oncol. 2021. PMID: 33407740 Free PMC article. Review.
-
Endoplasmic Reticulum Calcium Pumps and Tumor Cell Differentiation.Int J Mol Sci. 2020 May 9;21(9):3351. doi: 10.3390/ijms21093351. Int J Mol Sci. 2020. PMID: 32397400 Free PMC article. Review.
-
Discovery of novel SERCA inhibitors by virtual screening of a large compound library.Eur J Med Chem. 2011 May;46(5):1512-23. doi: 10.1016/j.ejmech.2011.01.069. Epub 2011 Feb 25. Eur J Med Chem. 2011. PMID: 21353727 Free PMC article.
-
Mesencephalic astrocyte-derived neurotrophic factor protects the heart from ischemic damage and is selectively secreted upon sarco/endoplasmic reticulum calcium depletion.J Biol Chem. 2012 Jul 27;287(31):25893-904. doi: 10.1074/jbc.M112.356345. Epub 2012 May 25. J Biol Chem. 2012. PMID: 22637475 Free PMC article.
-
Heck- and Suzuki-coupling approaches to novel hydroquinone inhibitors of calcium ATPase.Beilstein J Org Chem. 2019 Apr 24;15:971-975. doi: 10.3762/bjoc.15.94. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 31164934 Free PMC article.
References
-
- Moller JV, Juul B, le Maire M. Biochim Biophys Acta. 1996;1286:1. - PubMed
-
- Denmeade SR, Isaacs JT. Cancer Biol Ther. 2005;4:14. - PubMed
-
- Denmeade SR, Jakobsen CM, Janssen S, Khan SR, Garrett ES, Lilja H, Christensen SB, Isaacs JT. J Natl Cancer Inst. 2003;95:990. - PubMed
-
- Isaacs J. BJU International. 2005;96:35. - PubMed
-
- Treiman M, Caspersen C, Christensen SB. Trends Pharmacol Sci. 1998;19:131. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information

