Comparison of somatostatin and corticotrophin-releasing hormone immunoreactivity in forebrain neurons projecting to taste-responsive and non-responsive regions of the parabrachial nucleus in rat
- PMID: 19699720
- PMCID: PMC2769563
- DOI: 10.1016/j.brainres.2009.08.038
Comparison of somatostatin and corticotrophin-releasing hormone immunoreactivity in forebrain neurons projecting to taste-responsive and non-responsive regions of the parabrachial nucleus in rat
Abstract
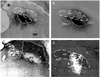
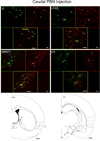
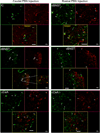
Several forebrain areas have been shown to project to the parabrachial nucleus (PBN) and exert inhibitory and excitatory influences on taste processing. The neurochemicals by which descending forebrain inputs modulate neural taste-evoked responses remain to be established. This study investigated the existence of somatostatin (SS) and corticotrophin-releasing factor (CRF) in forebrain neurons that project to caudal regions of the PBN responsive to chemical stimulation of the anterior tongue as well as more rostral unresponsive regions. Retrograde tracer was iontophoretically or pressure ejected from glass micropipettes, and 7 days later the animals were euthanized for subsequent immunohistochemical processing for co-localization of tracer with SS and CRF in tissue sections containing the lateral hypothalamus (LH), central nucleus of the amygdala (CeA), bed nucleus of the stria terminalis (BNST), and insular cortex (IC). In each forebrain site, robust labeling of cells with distinguishable nuclei and short processes was observed for SS and CRF. The results indicate that CRF neurons in each forebrain site send projections throughout the rostral caudal extent of the PBN with a greater percentage terminating in regions rostral to the anterior tongue-responsive area. For SS, the percentage of double-labeled neurons was more forebrain site specific in that only BNST and CeA exhibited significant numbers of double-labeled neurons. Few retrogradely labeled cells in LH co-expressed SS, while no double-labeled cells were observed in IC. Again, tracer injections into rostral PBN resulted in a greater percentage of double-labeled neurons in BNST and CeA compared to caudal injections. The present results suggest that some sources of descending forebrain input might utilize somatostatin and/or CRF to exert a broad influence on sensory information processing in the PBN.
Figures



References
-
- az-Casares A, Lopez-Gonzalez MV, Peinado-Aragones CA, Lara JP, Gonzalez-Baron S, wid-Milner MS. Role of the parabrachial complex in the cardiorespiratory response evoked from hypothalamic defense area stimulation in the anesthetized rat. Brain Res. 2009 Jul 7;1279:58–70. - PubMed
-
- Baird JP, Travers JB, Travers SP. Parametric analysis of gastric distension responses in the parabrachial nucleus. Am.J.Physiol Regul.Integr.Comp Physiol. 2001a;281(5):R1568–R1580. - PubMed
-
- Baird JP, Travers SP, Travers JB. Integration of gastric distension and gustatory responses in the parabrachial nucleus. Am.J.Physiol Regul.Integr.Comp Physiol. 2001b;281(5):R1581–R1593. - PubMed
-
- Bester H, Chapman V, Besson JM, Bernard JF. Physiological properties of the lamina I spinoparabrachial neurons in the rat. J.Neurophysiol. 2000;83(4):2239–2259. - PubMed
-
- Bester H, Menendez L, Besson JM, Bernard JF. Spino (trigemino) parabrachiohypothalamic pathway: electrophysiological evidence for an involvement in pain processes. J.Neurophysiol. 1995;73(2):568–585. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

