Microchimerism is strongly correlated with tolerance to noninherited maternal antigens in mice
- PMID: 19700665
- PMCID: PMC2766676
- DOI: 10.1182/blood-2009-03-213561
Microchimerism is strongly correlated with tolerance to noninherited maternal antigens in mice
Abstract
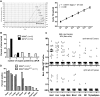
In mice and humans, the immunologic effects of developmental exposure to noninherited maternal antigens (NIMAs) are quite variable. This heterogeneity likely reflects differences in the relative levels of NIMA-specific T regulatory (T(R)) versus T effector (T(E)) cells. We hypothesized that maintenance of NIMA-specific T(R) cells in the adult requires continuous exposure to maternal cells and antigens (eg, maternal microchimerism [MMc]). To test this idea, we used 2 sensitive quantitative polymerase chain reaction (qPCR) tests to detect MMc in different organs of NIMA(d)-exposed H2(b) mice. MMc was detected in 100% of neonates and a majority (61%) of adults; nursing by a NIMA+ mother was essential for preserving MMc into adulthood. MMc was most prevalent in heart, lungs, liver, and blood, but was rarely detected in unfractionated lymphoid tissues. However, MMc was detectable in isolated CD4+, CD11b+, and CD11c+ cell subsets of spleen, and in lineage-positive cells in heart. Suppression of delayed type hypersensitivity (DTH) and in vivo lymphoproliferation correlated with MMc levels, suggesting a link between T(R) and maternal cell engraftment. In the absence of neonatal exposure to NIMA via breastfeeding, MMc was lost, which was accompanied by sensitization to NIMA in some offspring, indicating a role of oral exposure in maintaining a favorable T(R) > T(E) balance.
Figures

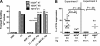



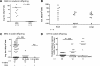
References
-
- Carstens J, Andersen HK, Spencer E, Madsen M. Cytomegalovirus infection in renal transplant recipients. Transpl Infect Dis. 2006;8:203–212. - PubMed
-
- López MM, Valenzuela JE, Alvarez FC, Lopez-Alvarez MR, Cecilia GS, Paricio PP. Long-term problems related to immunosuppression. Transpl Immunol. 2006;17:31–35. - PubMed
-
- Penn I. Post-transplant malignancy: the role of immunosuppression. Drug Saf. 2000;23:101–113. - PubMed
-
- Burlingham WJ, Grailer AP, Heisey DM, et al. The effect of tolerance to noninherited maternal HLA antigens on the survival of renal transplants from sibling donors. N Engl J Med. 1998;339:1657–1664. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

