A kinetic assessment of the C. elegans amyloid disaggregation activity enables uncoupling of disassembly and proteolysis
- PMID: 19701939
- PMCID: PMC2788278
- DOI: 10.1002/pro.234
A kinetic assessment of the C. elegans amyloid disaggregation activity enables uncoupling of disassembly and proteolysis
Erratum in
- Protein Sci. 2013 Nov;22(11):1688
Abstract
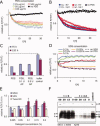
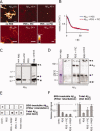
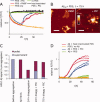
Protein aggregation is a common feature of late onset neurodegenerative disorders, including Alzheimer's disease. In Alzheimer's disease, misassembly of the Abeta peptide is genetically linked to proteotoxicity associated with disease etiology. A reduction in Abeta proteotoxicity is accomplished, in part, by the previously reported Abeta disaggregation and proteolysis activities-under partial control of heat shock factor 1, a transcription factor regulating proteostasis in the cytosol and negatively regulated by insulin growth factor signaling. Herein, we report an improved in vitro assay to quantify recombinant fibrillar Abeta disaggregation kinetics accomplished by the exogenous application of C.elegans extracts. With this assay we demonstrate that the Abeta disaggregation and proteolysis activities of C.elegans are separable. The disaggregation activity found in C.elegans preparations is more heat resistant than the proteolytic activity. Abeta disaggregation in the absence of proteolysis was found to be a reversible process. Future discovery of the molecular basis of the disaggregation and proteolysis activities offers the promise of delaying the age-onset proteotoxicity that leads to neurodegeneration in a spectrum of maladies.
Figures

References
-
- Kelly JW. The alternative conformations of amyloidogenic proteins and their multi-step assembly pathways. Curr Opin Struct Biol. 1998;8:101–106. - PubMed
-
- Dobson CM. Protein folding and misfolding. Nature. 2003;426:884–890. - PubMed
-
- Selkoe DJ. Cell biology of protein misfolding: the examples of Alzheimer's and Parkinson's diseases. Nat Cell Biol. 2004;6:1054–1061. - PubMed
-
- Sekijima Y, Wiseman RL, Matteson J, Hammarstrom P, Miller SR, Sawkar AR, Balch WE, Kelly JW. The biological and chemical basis for tissue-selective amyloid disease. Cell. 2005;121:73–85. - PubMed
-
- Scherzinger E, Lurz R, Turmaine M, Mangiarini L, Hollenbach B, Hasenbank R, Bates GP, Davies SW, Lehrach H, Wanker EE. Huntingtin-encoded polyglutamine expansions form amyloid-like protein aggregates in vitro and in vivo. Cell. 1997;90:549–558. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

