Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo
- PMID: 19710168
- PMCID: PMC2739140
- DOI: 10.1242/dev.038893
Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo
Abstract
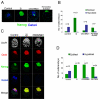
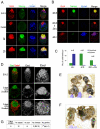
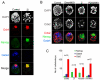
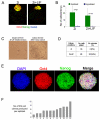
Embryonic stem (ES) cells can be derived and propagated from multiple strains of mouse and rat through application of small-molecule inhibitors of the fibroblast growth factor (FGF)/Erk pathway and of glycogen synthase kinase 3. These conditions shield pluripotent cells from differentiation-inducing stimuli. We investigate the effect of these inhibitors on the development of pluripotent epiblast in intact pre-implantation embryos. We find that blockade of Erk signalling from the 8-cell stage does not impede blastocyst formation but suppresses development of the hypoblast. The size of the inner cell mass (ICM) compartment is not reduced, however. Throughout the ICM, the epiblast-specific marker Nanog is expressed, and in XX embryos epigenetic silencing of the paternal X chromosome is erased. Epiblast identity and pluripotency were confirmed by contribution to chimaeras with germline transmission. These observations indicate that segregation of hypoblast from the bipotent ICM is dependent on FGF/Erk signalling and that in the absence of this signal, the entire ICM can acquire pluripotency. Furthermore, the epiblast does not require paracrine support from the hypoblast. Thus, naïve epiblast and ES cells are in a similar ground state, with an autonomous capacity for survival and replication, and high vulnerability to Erk signalling. We probed directly the relationship between naïve epiblast and ES cells. Dissociated ICM cells from freshly harvested late blastocysts gave rise to up to 12 ES cell clones per embryo when plated in the presence of inhibitors. We propose that ES cells are not a tissue culture creation, but are essentially identical to pre-implantation epiblast cells.
Figures
References
-
- Batlle-Morera, L., Smith, A. and Nichols, J. (2008). Parameters influencing derivation of embryonic stem cells from murine embryos. Genesis 46, 758-767. - PubMed
-
- Bradley, A., Evans, M. J., Kaufman, M. H. and Robertson, E. (1984). Formation of germ-line chimaeras from embryo-derived teratocarcinoma cell lines. Nature 309, 255-256. - PubMed
-
- Brons, I. G., Smithers, L. E., Trotter, M. W., Rugg-Gunn, P., Sun, B., Chuva de Sousa Lopes, S. M., Howlett, S. K., Clarkson, A., Ahrlund-Richter, L., Pedersen, R. A. et al. (2007). Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191-195. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

