Spodoptera frugiperda X-tox protein, an immune related defensin rosary, has lost the function of ancestral defensins
- PMID: 19710910
- PMCID: PMC2728511
- DOI: 10.1371/journal.pone.0006795
Spodoptera frugiperda X-tox protein, an immune related defensin rosary, has lost the function of ancestral defensins
Abstract
Background: X-tox proteins are a family of immune-related proteins only found in Lepidoptera and characterized by imperfectly conserved tandem repeats of several defensin-like motifs. Previous phylogenetic analysis of X-tox genes supported the hypothesis that X-tox have evolved from defensins in a lineage-specific gene evolution restricted to Lepidoptera. In this paper, we performed a protein study in which we asked whether X-tox proteins have conserved the antimicrobial functions of their ancestral defensins and have evolved as defensin reservoirs.
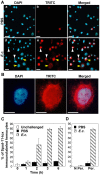
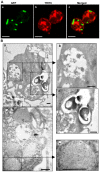
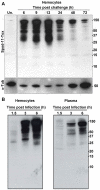
Methodology/principal findings: We followed the outcome of Spod-11-tox, an X-tox protein characterized in Spodoptera frugiperda, in bacteria-challenged larvae using both immunochemistry and antimicrobial assays. Three hours post infection, the Spod-11-tox protein was expressed in 80% of the two main classes of circulating hemocytes (granulocytes and plasmatocytes). Located in secretory granules of hemocytes, Spod-11-tox was never observed in contact with microorganisms entrapped within phagolyzosomes showing that Spod-11-tox is not involved in intracellular pathogen killing. In fact, the Spod-11-tox protein was found to be secreted into the hemolymph of experimentally challenged larvae. In order to determine antimicrobial properties of the Spod-11-tox protein, it was consequently fractionated according to a protocol frequently used for antimicrobial peptide purification. Over the course of purification, the anti-Spod-11-tox immunoreactivity was found to be dissociated from the antimicrobial activity. This indicates that Spod-11-tox is not processed into bioactive defensins in response to a microbial challenge.
Conclusions/significance: Altogether, our results show that X-tox proteins have not evolved as defensin reservoirs and have lost the antimicrobial properties of the ancestral insect defensins. The lepidopteran X-tox protein family will provide a valuable and tractable model to improve our knowledge on the molecular evolution of defensins, a class of innate immune effectors largely distributed over the three eukaryotic kingdoms.
Conflict of interest statement
Figures



Similar articles
-
X-tox: an atypical defensin derived family of immune-related proteins specific to Lepidoptera.Dev Comp Immunol. 2008;32(5):575-84. doi: 10.1016/j.dci.2007.09.004. Epub 2007 Oct 12. Dev Comp Immunol. 2008. PMID: 17988734
-
Evolutionary history of x-tox genes in three lepidopteran species: origin, evolution of primary and secondary structure and alternative splicing, generating a repertoire of immune-related proteins.Insect Biochem Mol Biol. 2013 Jan;43(1):54-64. doi: 10.1016/j.ibmb.2012.10.012. Epub 2012 Nov 9. Insect Biochem Mol Biol. 2013. PMID: 23142192
-
Identification, phylogenetic analysis and expression profile of an anionic insect defensin gene, with antibacterial activity, from bacterial-challenged cotton leafworm, Spodoptera littoralis.BMC Mol Biol. 2011 Nov 9;12:47. doi: 10.1186/1471-2199-12-47. BMC Mol Biol. 2011. PMID: 22067477 Free PMC article.
-
Defensins.Curr Opin Immunol. 1994 Aug;6(4):584-9. doi: 10.1016/0952-7915(94)90145-7. Curr Opin Immunol. 1994. PMID: 7946046 Review.
-
The evolution, function and mechanisms of action for plant defensins.Semin Cell Dev Biol. 2019 Apr;88:107-118. doi: 10.1016/j.semcdb.2018.02.004. Epub 2018 Feb 23. Semin Cell Dev Biol. 2019. PMID: 29432955 Review.
Cited by
-
Spodoptera frugiperda transcriptional response to infestation by Steinernema carpocapsae.Sci Rep. 2019 Sep 9;9(1):12879. doi: 10.1038/s41598-019-49410-8. Sci Rep. 2019. PMID: 31501491 Free PMC article.
-
RNA-Seq Study of Microbially Induced Hemocyte Transcripts from Larval Heliothis virescens (Lepidoptera: Noctuidae).Insects. 2012 Aug 14;3(3):743-62. doi: 10.3390/insects3030743. Insects. 2012. PMID: 26466627 Free PMC article.
-
Physiological Tradeoffs of Immune Response Differs by Infection Type in Pieris napi.Front Physiol. 2021 Jan 13;11:576797. doi: 10.3389/fphys.2020.576797. eCollection 2020. Front Physiol. 2021. PMID: 33519499 Free PMC article.
-
Functional analysis of four processing products from multiple precursors encoded by a lebocin-related gene from Manduca sexta.Dev Comp Immunol. 2010 Jun;34(6):638-47. doi: 10.1016/j.dci.2010.01.008. Epub 2010 Jan 28. Dev Comp Immunol. 2010. PMID: 20096726 Free PMC article.
-
Can insects develop resistance to insect pathogenic fungi?PLoS One. 2013;8(4):e60248. doi: 10.1371/journal.pone.0060248. Epub 2013 Apr 1. PLoS One. 2013. PMID: 23560083 Free PMC article.
References
-
- Ferrandon D, Imler JL, Hetru C, Hoffmann JA. The Drosophila systemic immune response: sensing and signalling during bacterial and fungal infections. Nat Rev Immunol. 2007;7:862–874. - PubMed
-
- Lemaitre B, Hoffmann J. The Host Defense of Drosophila melanogaster. Annu Rev Immunol 2007 - PubMed
-
- Tanaka H, Ishibashi J, Fujita K, Nakajima Y, Sagisaka A, et al. A genome-wide analysis of genes and gene families involved in innate immunity of Bombyx mori. Insect Biochem Mol Biol 2008 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

