Mammalian MagT1 and TUSC3 are required for cellular magnesium uptake and vertebrate embryonic development
- PMID: 19717468
- PMCID: PMC2732712
- DOI: 10.1073/pnas.0908332106
Mammalian MagT1 and TUSC3 are required for cellular magnesium uptake and vertebrate embryonic development
Abstract
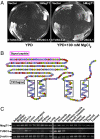
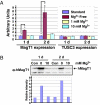
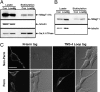
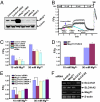
Magnesium (Mg(2+)) is the second most abundant cation in cells, yet relatively few mechanisms have been identified that regulate cellular levels of this ion. The most clearly identified Mg(2+) transporters are in bacteria and yeast. Here, we use a yeast complementary screen to identify two mammalian genes, MagT1 and TUSC3, as major mechanisms of Mg(2+) influx. MagT1 is universally expressed in all human tissues and its expression level is up-regulated in low extracellular Mg(2+). Knockdown of either MagT1 or TUSC3 protein significantly lowers the total and free intracellular Mg(2+) concentrations in mammalian cell lines. Morpholino knockdown of MagT1 and TUSC3 protein expression in zebrafish embryos results in early developmental arrest; excess Mg(2+) or supplementation with mammalian mRNAs can rescue the effects. We conclude that MagT1 and TUSC3 are indispensable members of the vertebrate plasma membrane Mg(2+) transport system.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Romani A. Regulation of magnesium homeostasis and transport in mammalian cells. Arch Biochem Biophys. 2007;458:90–102. - PubMed
-
- Fatholahi M, LaNoue K, Romani A, Scarpa A. Relationship between total and free cellular Mg(2+) during metabolic stimulation of rat cardiac myocytes and perfused hearts. Arch Biochem Biophys. 2000;374:395–401. - PubMed
-
- Harman AW, Nieminen AL, Lemasters JJ, Herman B. Cytosolic free magnesium, ATP and blebbing during chemical hypoxia in cultured rat hepatocytes. Biochem Biophys Res Commun. 1990;170:477–483. - PubMed
-
- Raju B, Murphy E, Levy LA, Hall RD, London RE. A fluorescent indicator for measuring cytosolic free magnesium. Am J Physiol. 1989;256:C540–C548. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

