Interferon regulatory factor-8 regulates bone metabolism by suppressing osteoclastogenesis
- PMID: 19718038
- PMCID: PMC2755267
- DOI: 10.1038/nm.2007
Interferon regulatory factor-8 regulates bone metabolism by suppressing osteoclastogenesis
Abstract
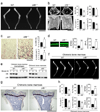
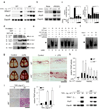
Bone metabolism results from a balance between osteoclast-driven bone resorption and osteoblast-mediated bone formation. Diseases such as periodontitis and rheumatoid arthritis are characterized by increased bone destruction due to enhanced osteoclastogenesis. Here we report that interferon regulatory factor-8 (IRF-8), a transcription factor expressed in immune cells, is a key regulatory molecule for osteoclastogenesis. IRF-8 expression in osteoclast precursors was downregulated during the initial phase of osteoclast differentiation induced by receptor activator of nuclear factor-kappaB ligand (RANKL), which is encoded by the Tnfsf11 gene. Mice deficient in Irf8 showed severe osteoporosis, owing to increased numbers of osteoclasts, and also showed enhanced bone destruction after lipopolysaccharide (LPS) administration. Irf8-/- osteoclast precursors underwent increased osteoclastogenesis in response to RANKL and tumor necrosis factor-alpha (TNF-alpha). IRF-8 suppressed osteoclastogenesis by inhibiting the function and expression of nuclear factor of activated T cells c1 (NFATc1). Our results show that IRF-8 inhibits osteoclast formation under physiological and pathological conditions and suggest a model where downregulation of inhibitory factors such as IRF-8 contributes to RANKL-mediated osteoclastogenesis.
Figures


References
-
- Wada T, Nakashima T, Hiroshi N, Penninger JM. RANKL-RANK signaling in osteoclastogenesis and bone disease. Trends Mol Med. 2006;12:17–25. - PubMed
-
- Lee SK, Lorenzo J. Cytokines regulating osteoclast formation and function. Curr Opin Rheumatol. 2006;18:411–418. - PubMed
-
- Takayanagi H, et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell. 2002;3:889–901. - PubMed
-
- Grigoriadis AE, et al. c-Fos: a key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science. 1994;266:443–448. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

