Donepezil improves episodic memory in young individuals vulnerable to the effects of sleep deprivation
- PMID: 19725251
- PMCID: PMC2717207
- DOI: 10.1093/sleep/32.8.999
Donepezil improves episodic memory in young individuals vulnerable to the effects of sleep deprivation
Abstract
Study objectives: We investigated if donepezil, a long-acting orally administered cholinesterase inhibitor, would reduce episodic memory deficits associated with 24 h of sleep deprivation.
Design: Double-blind, placebo-controlled, crossover study involving 7 laboratory visits over 2 months. Participants underwent 4 functional MRI scans; 2 sessions (donepezil or placebo) followed a normal night's sleep, and 2 sessions followed a night of sleep deprivation.
Setting: The study took place in a research laboratory.
Participants: 26 young, healthy volunteers with no history of any sleep, psychiatric, or neurologic disorders.
Interventions: 5 mg of donepezil was taken once daily for approximately 17 days.
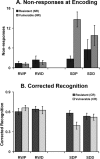
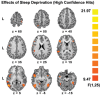
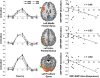
Measurements and results: Subjects were scanned while performing a semantic judgment task and tested for word recognition outside the scanner 45 minutes later. Sleep deprivation increased the frequency of non-responses at encoding and impaired delayed recognition. No benefit of donepezil was evident when participants were well rested. When sleep deprived, individuals who showed greater performance decline improved with donepezil, whereas more resistant individuals did not benefit. Accompanying these behavioral effects, there was corresponding modulation of task-related activation in functionally relevant brain regions. Brain regions identified in relation to donepezil-induced alteration in non-response rates could be distinguished from regions relating to improved recognition memory. This suggests that donepezil can improve delayed recognition in sleep-deprived persons by improving attention as well as enhancing memory encoding.
Conclusions: Donepezil reduced decline in recognition performance in individuals vulnerable to the effects of sleep deprivation. Additionally, our findings demonstrate the utility of combined fMRI-behavior evaluation in psychopharmacological studies.
Figures






Similar articles
-
Effects of donepezil on verbal memory after semantic processing in healthy older adults.Cogn Behav Neurol. 2008 Jun;21(2):57-64. doi: 10.1097/WNN.0b013e3181799df1. Cogn Behav Neurol. 2008. PMID: 18541979 Clinical Trial.
-
Effects of donepezil on cognitive performance after sleep deprivation.Hum Psychopharmacol. 2011 Dec;26(8):578-87. doi: 10.1002/hup.1248. Epub 2011 Dec 8. Hum Psychopharmacol. 2011. PMID: 22161694 Clinical Trial.
-
Cholinergic enhancement of episodic memory in healthy young adults.Psychopharmacology (Berl). 2005 Oct;182(1):170-9. doi: 10.1007/s00213-005-0043-2. Epub 2005 Sep 29. Psychopharmacology (Berl). 2005. PMID: 16021483 Clinical Trial.
-
The pharmacology of donepezil: a new treatment of Alzheimer's disease.Expert Opin Pharmacother. 1999 Nov;1(1):121-35. doi: 10.1517/14656566.1.1.121. Expert Opin Pharmacother. 1999. PMID: 11249555 Review.
-
A 10-year perspective on donepezil.Expert Opin Pharmacother. 2013 Feb;14(3):331-8. doi: 10.1517/14656566.2013.760543. Epub 2013 Jan 14. Expert Opin Pharmacother. 2013. PMID: 23316713 Review.
Cited by
-
Altered sigma-1 receptor expression in two animal models of cognitive impairment.Mol Imaging Biol. 2015 Apr;17(2):231-8. doi: 10.1007/s11307-014-0780-x. Mol Imaging Biol. 2015. PMID: 25273321
-
The rationale for consuming cognitive enhancement drugs in university students and teachers.PLoS One. 2013 Jul 17;8(7):e68821. doi: 10.1371/journal.pone.0068821. Print 2013. PLoS One. 2013. PMID: 23874778 Free PMC article.
-
Circadian rhythms, sleep deprivation, and human performance.Prog Mol Biol Transl Sci. 2013;119:155-90. doi: 10.1016/B978-0-12-396971-2.00007-5. Prog Mol Biol Transl Sci. 2013. PMID: 23899598 Free PMC article. Review.
-
Network dynamics predict improvement in working memory performance following donepezil administration in healthy young adults.Neuroimage. 2014 Mar;88:228-41. doi: 10.1016/j.neuroimage.2013.11.020. Epub 2013 Nov 21. Neuroimage. 2014. PMID: 24269569 Free PMC article.
-
Concordance of multiple methods to define resiliency and vulnerability to sleep loss depends on Psychomotor Vigilance Test metric.Sleep. 2022 Jan 11;45(1):zsab249. doi: 10.1093/sleep/zsab249. Sleep. 2022. PMID: 34624897 Free PMC article.
References
-
- Born J, Rasch B, Gais S. Sleep to remember. Neuroscientist. 2006;12:410–24. - PubMed
-
- Maquet P. The role of sleep in learning and memory. Science. 2001;294:1048–52. - PubMed
-
- Walker MP, Stickgold R. Sleep, memory, and plasticity. Annu Rev Psychol. 2006;57:139–66. - PubMed
-
- Chee MW, Chuah LY. Functional neuroimaging insights into how sleep and sleep deprivation affect memory and cognition. Curr Opin Neurol. 2008;21:417–23. - PubMed
-
- Bonnet MH, Balkin TJ, Dinges DF, Roehrs T, Rogers NL, Wesensten NJ. The use of stimulants to modify performance during sleep loss: a review by the sleep deprivation and Stimulant Task Force of the American Academy of Sleep Medicine. Sleep. 2005;28:1163–87. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

