Evidence of inability of human cytomegalovirus to reactivate Kaposi's sarcoma-associated herpesvirus from latency in body cavity-based lymphocytes
- PMID: 19726225
- PMCID: PMC2789304
- DOI: 10.1016/j.jcv.2009.07.025
Evidence of inability of human cytomegalovirus to reactivate Kaposi's sarcoma-associated herpesvirus from latency in body cavity-based lymphocytes
Abstract
Background: Kaposi's sarcoma-associated herpesvirus (KSHV; also known as human herpesvirus 8 (HHV-8)) has been determined to be the most frequent cause of malignancies in AIDS patients. It is associated primarily with Kaposi's sarcoma (KS) and primary effusion lymphoma (PEL), as well as with multicentric Castleman's disease (MCD).(2) The switch from the latent to the lytic stage is important in the maintenance of malignancy and viral infection. So far, the mechanism of its reactivation has not been fully understood.
Objectives: Human cytomegalovirus (HCMV) and KSHV might infect the same cells, and it was found by other groups that several viruses could reactivate KSHV from latency. We investigate whether HCMV infection could reactivate KSHV from latency in body cavity-based lymphocyte (BCBL-1) cells.
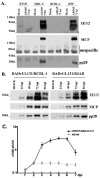
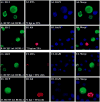
Study design and results: Laboratory strains of HCMV cannot infect B cells. In this article, we demonstrate that the UL131-repaired HCMV (vDW215-BADrUL131) derived from AD169 strain is able to infect B lymphocytes. We directly infected KSHV latent cells including BCBL-1 with vDW215-BADrUL131 to evaluate the ability of HCMV to reactivate KSHV. Inconsistent with previous reports in human fibroblast cells, our results provide direct evidence that HCMV is unable to reactivate KSHV from latency-to-lytic infection in BCBL-1 cell lines. As a control, herpes simplex virus type 1 (HSV-1) was shown to be able to reactivate KSHV.
Conclusions: Our observations, different from others, suggest that reactivation mechanisms for KSHV might vary in different cells.
Conflict of interest statement
Conflicts of Interest Statements
1 We don’t have any conflict interests regarding this paper.
Figures

Similar articles
-
Nuclear Innate Immune DNA Sensor IFI16 Is Degraded during Lytic Reactivation of Kaposi's Sarcoma-Associated Herpesvirus (KSHV): Role of IFI16 in Maintenance of KSHV Latency.J Virol. 2016 Sep 12;90(19):8822-41. doi: 10.1128/JVI.01003-16. Print 2016 Oct 1. J Virol. 2016. PMID: 27466416 Free PMC article.
-
Activation of Kaposi's sarcoma-associated herpesvirus (human herpesvirus 8) lytic replication by human cytomegalovirus.J Virol. 2001 Feb;75(3):1378-86. doi: 10.1128/JVI.75.3.1378-1386.2001. J Virol. 2001. PMID: 11152511 Free PMC article.
-
Establishment and maintenance of Kaposi's sarcoma-associated herpesvirus latency in B cells.J Virol. 2005 Nov;79(22):14383-91. doi: 10.1128/JVI.79.22.14383-14391.2005. J Virol. 2005. PMID: 16254372 Free PMC article.
-
Regulation of KSHV Latency and Lytic Reactivation.Viruses. 2020 Sep 17;12(9):1034. doi: 10.3390/v12091034. Viruses. 2020. PMID: 32957532 Free PMC article. Review.
-
KSHV Genome Replication and Maintenance in Latency.Adv Exp Med Biol. 2018;1045:299-320. doi: 10.1007/978-981-10-7230-7_14. Adv Exp Med Biol. 2018. PMID: 29896673 Review.
Cited by
-
Functional interaction of nuclear domain 10 and its components with cytomegalovirus after infections: cross-species host cells versus native cells.PLoS One. 2011 Apr 28;6(4):e19187. doi: 10.1371/journal.pone.0019187. PLoS One. 2011. PMID: 21552525 Free PMC article.
-
Sirtuin 6 Attenuates Kaposi's Sarcoma-Associated Herpesvirus Reactivation by Suppressing Ori-Lyt Activity and Expression of RTA.J Virol. 2019 Mar 21;93(7):e02200-18. doi: 10.1128/JVI.02200-18. Print 2019 Apr 1. J Virol. 2019. PMID: 30651359 Free PMC article.
-
The Impact of Co-Infections for Human Gammaherpesvirus Infection and Associated Pathologies.Int J Mol Sci. 2023 Aug 22;24(17):13066. doi: 10.3390/ijms241713066. Int J Mol Sci. 2023. PMID: 37685871 Free PMC article. Review.
-
Cellular microRNAs 498 and 320d regulate herpes simplex virus 1 induction of Kaposi's sarcoma-associated herpesvirus lytic replication by targeting RTA.PLoS One. 2013;8(2):e55832. doi: 10.1371/journal.pone.0055832. Epub 2013 Feb 13. PLoS One. 2013. PMID: 23418466 Free PMC article.
References
-
- Akter P, Cunningham C, McSharry BP, Dolan A, Addison C, Dargan DJ, Hassan-Walker AF, Emery VC, Griffiths PD, Wilkinson GW, Davison AJ. Two novel spliced genes in human cytomegalovirus. J Gen Virol. 2003;84:1117–22. - PubMed
-
- Ambroziak JA, Blackbourn DJ, Herndier BG, Glogau RG, Gullett JH, McDonald AR, Lennette ET, Levy JA. Herpes-like sequences in HIV-infected and uninfected Kaposi’s sarcoma patients. Science. 1995;268:582–3. - PubMed
-
- Atula T, Grenman R, Klemi P, Syrjanen S. Human papillomavirus, Epstein-Barr virus, human herpesvirus 8 and human cytomegalovirus involvement in salivary gland tumours. Oral Oncol. 1998;34:391–5. - PubMed
-
- Boshoff C, Whitby D, Talbot S, Weiss RA. Etiology of AIDS-related Kaposi’s sarcoma and lymphoma. Oral Dis. 1997;3 Suppl 1:S129–32. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

