The AKV murine leukemia virus is restricted and hypermutated by mouse APOBEC3
- PMID: 19726503
- PMCID: PMC2772689
- DOI: 10.1128/JVI.01430-09
The AKV murine leukemia virus is restricted and hypermutated by mouse APOBEC3
Abstract
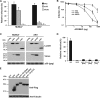
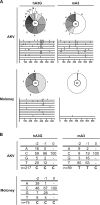
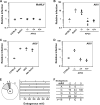
APOBEC3 proteins are potent restriction factors against retroviral infection in primates. This restriction is accompanied by hypermutations in the retroviral genome that are attributable to the cytidine deaminase activity of the APOBEC3 proteins. Studies of nucleotide sequence diversity among endogenous gammaretroviruses suggest that the evolution of endogenous retroelements could have been shaped by the mutagenic cytidine deaminase activity of APOBEC3. In mice, however, APOBEC3 appears to restrict exogenous murine retroviruses in the absence of detectable levels of deamination. AKV is an endogenous retrovirus that is involved in causing a high incidence of thymic lymphoma in AKR mice. A comparative analysis of several mouse strains revealed a relatively low level of APOBEC3 expression in AKR mice. Here we show that endogenous mouse APOBEC3 restricts AKV infection and that this restriction likely reflects polymorphisms affecting APOBEC3 abundance rather than differences in the APOBEC3 isoforms expressed. We also observe that restriction of AKV by APOBEC3 is accompanied by G-->A hypermutations in the viral genome. Our findings demonstrate that APOBEC3 acts as a restriction factor in rodents affecting the strain tropism of AKV, and they provide good support for the proposal that APOBEC3-mediated hypermutation contributed to the evolution of endogenous rodent retroviral genomes.
Figures


Similar articles
-
Deaminase-Dead Mouse APOBEC3 Is an In Vivo Retroviral Restriction Factor.J Virol. 2018 May 14;92(11):e00168-18. doi: 10.1128/JVI.00168-18. Print 2018 Jun 1. J Virol. 2018. PMID: 29593034 Free PMC article.
-
Mouse APOBEC3 expression in NIH 3T3 cells mediates hypermutation of AKV murine leukemia virus.Virology. 2018 May;518:377-384. doi: 10.1016/j.virol.2018.03.014. Epub 2018 Mar 30. Virology. 2018. PMID: 29605684 Free PMC article.
-
The glycosylated Gag protein of a murine leukemia virus inhibits the antiretroviral function of APOBEC3.J Virol. 2010 Oct;84(20):10933-6. doi: 10.1128/JVI.01023-10. Epub 2010 Aug 11. J Virol. 2010. PMID: 20702647 Free PMC article.
-
Mouse APOBEC3 Restriction of Retroviruses.Viruses. 2020 Oct 27;12(11):1217. doi: 10.3390/v12111217. Viruses. 2020. PMID: 33121095 Free PMC article. Review.
-
[Molecular evolution of physiologically functioning anti-retroviral APOBEC3 deaminases].Uirusu. 2012 Jun;62(1):27-38. doi: 10.2222/jsv.62.27. Uirusu. 2012. PMID: 23189822 Review. Japanese.
Cited by
-
Footprint of APOBEC3 on the genome of human retroelements.J Virol. 2013 Jul;87(14):8195-204. doi: 10.1128/JVI.00298-13. Epub 2013 May 22. J Virol. 2013. PMID: 23698293 Free PMC article.
-
Immunoglobulin VH gene diversity and somatic hypermutation during SIV infection of rhesus macaques.Immunogenetics. 2015 Jul;67(7):355-70. doi: 10.1007/s00251-015-0844-3. Epub 2015 May 21. Immunogenetics. 2015. PMID: 25994147 Free PMC article.
-
Mouse APOBEC3 interferes with autocatalytic cleavage of murine leukemia virus Pr180gag-pol precursor and inhibits Pr65gag processing.PLoS Pathog. 2019 Dec 12;15(12):e1008173. doi: 10.1371/journal.ppat.1008173. eCollection 2019 Dec. PLoS Pathog. 2019. PMID: 31830125 Free PMC article.
-
Restriction of porcine endogenous retrovirus by porcine APOBEC3 cytidine deaminases.J Virol. 2011 Apr;85(8):3842-57. doi: 10.1128/JVI.01880-10. Epub 2011 Feb 9. J Virol. 2011. PMID: 21307203 Free PMC article.
-
Reassessment of murine APOBEC1 as a retrovirus restriction factor in vivo.Virology. 2014 Nov;468-470:601-608. doi: 10.1016/j.virol.2014.09.006. Epub 2014 Oct 7. Virology. 2014. PMID: 25303118 Free PMC article.
References
-
- Aagaard, L., J. G. Mikkelsen, S. Warming, M. Duch, and F. S. Pedersen. 2002. Fv1-like restriction of N-tropic replication-competent murine leukaemia viruses in mCAT-1-expressing human cells. J. Gen. Virol. 83:439-442. - PubMed
-
- Abudu, A., A. Takaori-Kondo, T. Izumi, K. Shirakawa, M. Kobayashi, A. Sasada, K. Fukunaga, and T. Uchiyama. 2006. Murine retrovirus escapes from murine APOBEC3 via two distinct novel mechanisms. Curr. Biol. 16:1565-1570. - PubMed
-
- Bachrach, E., M. Pelegrin, M. Piechaczyk, F. S. Pedersen, and M. Duch. 2002. Efficient gene transfer into spleen cells of newborn mice by a replication-competent retroviral vector. Virology 293:328-334. - PubMed
-
- Beale, R. C., S. K. Petersen-Mahrt, I. N. Watt, R. S. Harris, C. Rada, and M. S. Neuberger. 2004. Comparison of the differential context-dependence of DNA deamination by APOBEC enzymes: correlation with mutation spectra in vivo. J. Mol. Biol. 337:585-596. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

