Regulation of VASP phosphorylation in cardiac myocytes: differential regulation by cyclic nucleotides and modulation of protein expression in diabetic and hypertrophic heart
- PMID: 19734360
- PMCID: PMC2781375
- DOI: 10.1152/ajpheart.00595.2009
Regulation of VASP phosphorylation in cardiac myocytes: differential regulation by cyclic nucleotides and modulation of protein expression in diabetic and hypertrophic heart
Abstract
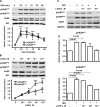
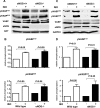
Vasodilator-stimulated phosphoprotein (VASP) is a major substrate for cyclic nucleotide-dependent kinases that has been implicated in cardiac pathology, yet many aspects of VASP's molecular regulation in cardiomyocytes are incompletely understood. In these studies, we explored the role of VASP, both in signaling pathways in isolated murine myocytes, as well as in a model of cardiac hypertrophy in VASP(null) mice. We found that the beta-adrenergic agonist isoproterenol promotes the rapid and reversible phosphorylation of VASP at Ser157 and Ser239. Forskolin and the cAMP analog 8-(4-chlorophenylthio)-cAMP promote a similar pattern of VASP phosphorylation at both sites. The effects of isoproterenol are blocked by atenolol and by compound H-89, an inhibitor of the cAMP-dependent protein kinase. By contrast, phosphorylation of VASP only at Ser239 is seen following activation of particulate guanylate cyclase by atrial natriuretic peptide, or following activation of soluble guanylate cyclase by sodium nitroprusside, or following treatment of myocytes with cGMP analog. We found that basal and isoproterenol-induced VASP phosphorylation is entirely unchanged in cardiomyocytes isolated from either endothelial or neuronal nitric oxide synthase knockout mice. In cardiomyocytes isolated from diabetic mice, only basal VASP phosphorylation is increased, whereas, in cells isolated from mice subjected to ascending aortic constriction (AAC), we found a significant increase in basal VASP expression, along with an increase in VASP phosphorylation, compared with cardiac myocytes isolated from sham-operated mice. Moreover, there is further increase in VASP phosphorylation in cells isolated from hypertrophic hearts following isoproterenol treatment. Finally, we found that VASP(null) mice subjected to transverse aortic constriction develop cardiac hypertrophy with a pattern similar to VASP(+/+) mice. Our findings establish differential receptor-modulated regulation of VASP phosphorylation in cardiomyocytes by cyclic nucleotides. Furthermore, these studies demonstrate for the first time that VASP expression is upregulated in hypertrophied heart.
Figures







Similar articles
-
Phosphorylation of blood vessel vasodilator-stimulated phosphoprotein at serine 239 as a functional biochemical marker of endothelial nitric oxide/cyclic GMP signaling.Mol Pharmacol. 2002 Feb;61(2):312-9. doi: 10.1124/mol.61.2.312. Mol Pharmacol. 2002. PMID: 11809855
-
Nitroxyl (HNO) stimulates soluble guanylyl cyclase to suppress cardiomyocyte hypertrophy and superoxide generation.PLoS One. 2012;7(4):e34892. doi: 10.1371/journal.pone.0034892. Epub 2012 Apr 10. PLoS One. 2012. PMID: 22506056 Free PMC article.
-
Effects of angiotensin II infusion on the expression and function of NAD(P)H oxidase and components of nitric oxide/cGMP signaling.Circ Res. 2002 Mar 8;90(4):E58-65. doi: 10.1161/01.res.0000012569.55432.02. Circ Res. 2002. PMID: 11884382
-
Regulation of phospholamban and troponin-I phosphorylation in the intact rat cardiomyocytes by adrenergic and cholinergic stimuli: roles of cyclic nucleotides, calcium, protein kinases and phosphatases and depolarization.Mol Cell Biochem. 1995 Aug-Sep;149-150:103-26. doi: 10.1007/BF01076569. Mol Cell Biochem. 1995. PMID: 8569720 Review.
-
Compartmentalization of β-adrenergic signals in cardiomyocytes.Trends Cardiovasc Med. 2013 Oct;23(7):250-6. doi: 10.1016/j.tcm.2013.02.001. Epub 2013 Mar 23. Trends Cardiovasc Med. 2013. PMID: 23528751 Free PMC article. Review.
Cited by
-
Upregulation of Myocardial and Vascular Phosphodiesterase 9A in A Model of Atherosclerotic Cardiovascular Disease.Int J Mol Sci. 2018 Sep 22;19(10):2882. doi: 10.3390/ijms19102882. Int J Mol Sci. 2018. PMID: 30249014 Free PMC article.
-
Thioredoxin reductase was nitrated in the aging heart after myocardial ischemia/reperfusion.Rejuvenation Res. 2013 Oct;16(5):377-85. doi: 10.1089/rej.2013.1437. Rejuvenation Res. 2013. PMID: 23802942 Free PMC article.
-
Stimulation of soluble guanylate cyclase improves donor organ function in rat heart transplantation.Sci Rep. 2020 Mar 24;10(1):5358. doi: 10.1038/s41598-020-62156-y. Sci Rep. 2020. PMID: 32210293 Free PMC article.
-
Cinaciguat prevents the development of pathologic hypertrophy in a rat model of left ventricular pressure overload.Sci Rep. 2016 Nov 17;6:37166. doi: 10.1038/srep37166. Sci Rep. 2016. PMID: 27853261 Free PMC article.
-
Insulin-dependent metabolic and inotropic responses in the heart are modulated by hydrogen peroxide from NADPH-oxidase isoforms NOX2 and NOX4.Free Radic Biol Med. 2017 Dec;113:16-25. doi: 10.1016/j.freeradbiomed.2017.09.006. Epub 2017 Sep 14. Free Radic Biol Med. 2017. PMID: 28917508 Free PMC article.
References
-
- An D, Rodriguez B. Role of changes in cardiac metabolism in development of diabetic cardiomyopathy. Am J Physiol Heart Circ Physiol 291: H1489–H1506, 2006 - PubMed
-
- Aszodi A, Pfeifer A, Ahmad M, Glauner M, Zhou XH, Ny L, Andersson KE, Kehrel B, Offermanns S, Fassler R. The vasodilator-stimulated phosphoprotein (VASP) is involved in cGMP- and cAMP-mediated inhibition of agonist-induced platelet aggregation, but is dispensable for smooth muscle function. EMBO J 18: 37–48, 1999 - PMC - PubMed
-
- Belhassen L, Feron O, Kaye DM, Michel T, Kelly RA. Regulation by cAMP of post-translational processing and subcellular targeting of endothelial nitric-oxide synthase (type 3) in cardiac myocytes. J Biol Chem 272: 11198–11204, 1997 - PubMed
-
- Blume C, Benz PM, Walter U, Ha J, Kemp BE, Renne T. AMP-activated protein kinase impairs endothelial actin cytoskeleton assembly by phosphorylating vasodilator-stimulated phosphoprotein. J Biol Chem 282: 4601–4612, 2007 - PubMed
-
- Brancaccio M, Hirsch E, Notte A, Selvetella G, Lembo G, Tarone G. Integrin signalling: the tug-of-war in heart hypertrophy. Cardiovasc Res 70: 422–433, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

