A directional Wnt/beta-catenin-Sox2-proneural pathway regulates the transition from proliferation to differentiation in the Xenopus retina
- PMID: 19736324
- PMCID: PMC2739145
- DOI: 10.1242/dev.040451
A directional Wnt/beta-catenin-Sox2-proneural pathway regulates the transition from proliferation to differentiation in the Xenopus retina
Abstract
Progenitor cells in the central nervous system must leave the cell cycle to become neurons and glia, but the signals that coordinate this transition remain largely unknown. We previously found that Wnt signaling, acting through Sox2, promotes neural competence in the Xenopus retina by activating proneural gene expression. We now report that Wnt and Sox2 inhibit neural differentiation through Notch activation. Independently of Sox2, Wnt stimulates retinal progenitor proliferation and this, when combined with the block on differentiation, maintains retinal progenitor fates. Feedback inhibition by Sox2 on Wnt signaling and by the proneural transcription factors on Sox2 mean that each element of the core pathway activates the next element and inhibits the previous one, providing a directional network that ensures retinal cells make the transition from progenitors to neurons and glia.
Figures




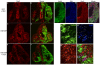
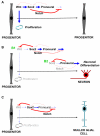
References
-
- Bani-Yaghoub, M., Tremblay, R. G., Lei, J. X., Zhang, D., Zurakowski, B., Sandhu, J. K., Smith, B., Ribecco-Lutkiewicz, M., Kennedy, J., Walker, P. R. et al. (2006). Role of Sox2 in the development of the mouse neocortex. Dev. Biol. 295, 52-66. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

