The DLK-1 kinase promotes mRNA stability and local translation in C. elegans synapses and axon regeneration
- PMID: 19737525
- PMCID: PMC2772821
- DOI: 10.1016/j.cell.2009.06.023
The DLK-1 kinase promotes mRNA stability and local translation in C. elegans synapses and axon regeneration
Abstract
Growth cone guidance and synaptic plasticity involve dynamic local changes in proteins at axons and dendrites. The Dual-Leucine zipper Kinase MAPKKK (DLK) has been previously implicated in synaptogenesis and axon outgrowth in C. elegans and other animals. Here we show that in C. elegans DLK-1 regulates not only proper synapse formation and axon morphology but also axon regeneration by influencing mRNA stability. DLK-1 kinase signals via a MAPKAP kinase, MAK-2, to stabilize the mRNA encoding CEBP-1, a bZip protein related to CCAAT/enhancer-binding proteins, via its 3'UTR. Inappropriate upregulation of cebp-1 in adult neurons disrupts synapses and axon morphology. CEBP-1 and the DLK-1 pathway are essential for axon regeneration after laser axotomy in adult neurons, and axotomy induces translation of CEBP-1 in axons. Our findings identify the DLK-1 pathway as a regulator of mRNA stability in synapse formation and maintenance and also in adult axon regeneration.
Figures


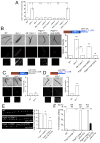


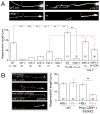
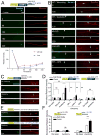
References
-
- Alberini CM, Ghirardi M, Metz R, Kandel ER. C/EBP is an immediate-early gene required for the consolidation of long-term facilitation in Aplysia. Cell. 1994;76:1099–1114. - PubMed
-
- Bertrand E, Chartrand P, Schaefer M, Shenoy SM, Singer RH, Long RM. Localization of ASH1 mRNA particles in living yeast. Mol Cell. 1998;2:437–445. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

