Stimulus-specific distinctions in spatial and temporal dynamics of stress-activated protein kinase kinase kinases revealed by a fluorescence resonance energy transfer biosensor
- PMID: 19737916
- PMCID: PMC2772575
- DOI: 10.1128/MCB.00571-09
Stimulus-specific distinctions in spatial and temporal dynamics of stress-activated protein kinase kinase kinases revealed by a fluorescence resonance energy transfer biosensor
Abstract
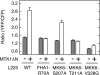
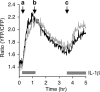
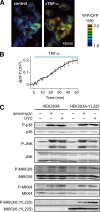
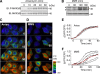
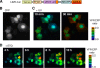
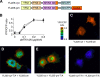
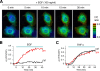
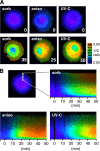
The stress-activated protein kinases (SAPKs), namely, p38 and JNK, are members of the mitogen-activated protein kinase family and are important determinants of cell fate when cells are exposed to environmental stresses such as UV and osmostress. SAPKs are activated by SAPK kinases (SAP2Ks), which are in turn activated by various SAP2K kinases (SAP3Ks). Because conventional methods, such as immunoblotting using phospho-specific antibodies, measure the average activity of SAP3Ks in a cell population, the intracellular dynamics of SAP3K activity are largely unknown. Here, we developed a reporter of SAP3K activity toward the MKK6 SAP2K, based on fluorescence resonance energy transfer, that can uncover the dynamic behavior of SAP3K activation in cells. Using this reporter, we demonstrated that SAP3K activation occurs either synchronously or asynchronously among a cell population and in different cellular compartments in single cells, depending on the type of stress applied. In particular, SAP3Ks are activated by epidermal growth factor and osmostress on the plasma membrane, by anisomycin and UV in the cytoplasm, and by etoposide in the nucleus. These observations revealed previously unknown heterogeneity in SAPK responses and supplied answers to the question of the cellular location in which various stresses induce stimulus-specific SAPK responses.
Figures



References
-
- Awad, M. M., H. Enslen, J. M. Boylan, R. J. Davis, and P. A. Gruppuso. 2000. Growth regulation via p38 mitogen-activated protein kinase in developing liver. J. Biol. Chem. 275:38716-38721. - PubMed
-
- Chen, Z., T. B. Gibson, F. Robinson, L. Silvestro, G. Pearson, B. Xu, A. Wright, C. Vanderbilt, and M. H. Cobb. 2001. MAP kinases. Chem. Rev. 101:2449-2476. - PubMed
-
- Dong, C., R. J. Davis, and R. A. Flavell. 2002. MAP kinases in the immune response. Annu. Rev. Immunol. 20:55-72. - PubMed
-
- Durocher, D., I. A. Taylor, D. Sarbassova, L. F. Haire, S. L. Westcott, S. P. Jackson, S. J. Smerdon, and M. B. Yaffe. 2000. The molecular basis of FHA domain: phosphopeptide binding domain specificity and implications for phospho-dependent signaling mechanisms. Mol. Cell 6:1169-1182. - PubMed
-
- Iordanov, M. S., D. Pribnow, J. L. Magun, T.-H. Dinh, J. A. Pearson, and B. E. Magun. 1998. Ultraviolet radiation triggers the ribotoxic stress response in mammalian cells. J. Biol. Chem. 273:15794-15803. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
