Interleukin-12 is sufficient to promote antigen-independent interferon-gamma production by CD8 T cells in old mice
- PMID: 19740329
- PMCID: PMC2753923
- DOI: 10.1111/j.1365-2567.2009.03061.x
Interleukin-12 is sufficient to promote antigen-independent interferon-gamma production by CD8 T cells in old mice
Abstract
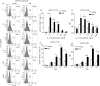
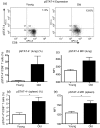
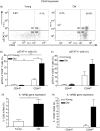
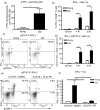
Numerous functional defects have been identified in naive T cells from aged mice, including deficiencies in proliferation, cytokine production and signal transduction. It is well documented that the ratio of naïve to memory T cells significantly decreases with age resulting in the majority of T cells from aged hosts expressing activated/memory T-cell markers (CD44(hi)), yet it is unclear whether T cells with a CD44(hi) phenotype in aged hosts are functionally equivalent to T cells with a similar phenotype in young hosts. We have identified a population of CD44(hi) CD8 T cells in old mice that are capable of secreting interferon-gamma (IFN-gamma) in response to interleukin-12 (IL-12) stimulation. This occurred in the absence of T-cell receptor engagement, a function that was not observed in CD8 T cells from young mice. This phenotype was associated with increased IL-12 receptor beta2 gene expression and IL-12 induced signal transducer and activator of transcription 4 (STAT-4) activation, even when CD8 T-cell numbers from young and old mice were normalized for CD44(hi) expression. Furthermore, we demonstrate that IL-12-induced STAT-4 activation was required for T helper type 1 (Th1) cytokine-induced IFN-gamma production in CD8 T cells. These data illustrate that old mice possess a specialized subset of CD44(hi) CD8 T cells with an enhanced responsiveness to IL-12, enabling these cells to produce substantial amounts of IFN-gamma in response to Th1 cytokine stimulation. We have therefore identified a functional difference in the populations of CD44(hi) CD8 T cells from young and old mice, and believe that understanding age-associated immunological changes is essential for helping the elderly combat deadly diseases.
Figures
Similar articles
-
Corrective effects of interleukin-12 on age-related deficiencies in IFN-gamma production and IL-12Rbeta2 expression in virus-specific CD8+ T cells.J Interferon Cytokine Res. 2000 Feb;20(2):235-45. doi: 10.1089/107999000312658. J Interferon Cytokine Res. 2000. PMID: 10714560
-
CD8 T cells in innate immune responses: using STAT4-dependent but antigen-independent pathways to gamma interferon during viral infection.mBio. 2014 Oct 21;5(5):e01978-14. doi: 10.1128/mBio.01978-14. mBio. 2014. PMID: 25336459 Free PMC article.
-
CD8+, but not CD4+ effector/memory T cells, express the CD44highCD45RBhigh phenotype with aging, which displays reduced expression levels of P2X7 receptor and ATP-induced cellular responses.FASEB J. 2019 Mar;33(3):3225-3236. doi: 10.1096/fj.201800867R. Epub 2018 Nov 1. FASEB J. 2019. PMID: 30383448
-
T helper differentiation proceeds through Stat1-dependent, Stat4-dependent and Stat4-independent phases.Curr Top Microbiol Immunol. 1999;238:13-26. doi: 10.1007/978-3-662-09709-0_2. Curr Top Microbiol Immunol. 1999. PMID: 10087648 Review.
-
Hiding in Plain Sight: Virtually Unrecognizable Memory Phenotype CD8+ T cells.Int J Mol Sci. 2020 Nov 16;21(22):8626. doi: 10.3390/ijms21228626. Int J Mol Sci. 2020. PMID: 33207648 Free PMC article. Review.
Cited by
-
Does tuberculosis threaten our ageing populations?BMC Infect Dis. 2016 Mar 11;16:119. doi: 10.1186/s12879-016-1451-0. BMC Infect Dis. 2016. PMID: 26968654 Free PMC article. Review.
-
T cell activation and IFNγ modulate organ dysfunction in LPS-mediated inflammation.J Leukoc Biol. 2022 Aug;112(2):221-232. doi: 10.1002/JLB.4HI0921-492R. Epub 2022 Feb 9. J Leukoc Biol. 2022. PMID: 35141943 Free PMC article.
-
The Impact of Aging on the Lung Alveolar Environment, Predetermining Susceptibility to Respiratory Infections.Front Aging. 2022 Jan 19;3:818700. doi: 10.3389/fragi.2022.818700. eCollection 2022. Front Aging. 2022. PMID: 35821836 Free PMC article. Review.
-
Effector Mechanisms of CD8+ HLA-DR+ T Cells in Breast Cancer Patients Who Respond to Neoadjuvant Chemotherapy.Cancers (Basel). 2021 Dec 7;13(24):6167. doi: 10.3390/cancers13246167. Cancers (Basel). 2021. PMID: 34944786 Free PMC article.
-
Identification of an Increased Alveolar Macrophage Subpopulation in Old Mice That Displays Unique Inflammatory Characteristics and Is Permissive to Mycobacterium tuberculosis Infection.J Immunol. 2019 Oct 15;203(8):2252-2264. doi: 10.4049/jimmunol.1900495. Epub 2019 Sep 11. J Immunol. 2019. PMID: 31511357 Free PMC article.
References
-
- Beers NH, Berkow R. The Merck Manual of Geriatrics. 3rd edn. Whitehouse Station, NJ: Merck Research Laboratories; 2000.
-
- Niederman MS, Fein AM. Pneumonia in the elderly. Clin Geriatr Med. 1986;2:241–68. - PubMed
-
- Orme IM. Aging and immunity to tuberculosis: increased susceptibility of old mice reflects a decreased capacity to generate mediator T lymphocytes. J Immunol. 1987;138:4414–8. - PubMed
-
- Stead WW, Dutt AK. Tuberculosis in elderly persons. Annu Rev Med. 1991;42:267–76. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

