A naturally occurring Rgg variant in serotype M3 Streptococcus pyogenes does not activate speB expression due to altered specificity of DNA binding
- PMID: 19752034
- PMCID: PMC2786450
- DOI: 10.1128/IAI.00373-09
A naturally occurring Rgg variant in serotype M3 Streptococcus pyogenes does not activate speB expression due to altered specificity of DNA binding
Abstract
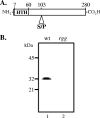
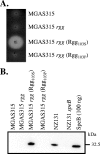
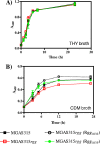
The transcriptional regulator Rgg of Streptococcus pyogenes is essential for expression of the secreted cysteine protease SpeB. Although all isolates of S. pyogenes possess the speB gene, not all of them produce the protein in vitro. In a murine model of infection, the absence of SpeB production is associated with invasive disease. We speculated that naturally occurring mutations in rgg, which would also abrogate SpeB production, may be present in invasive isolates of S. pyogenes. Examination of the inferred Rgg sequences available in public databases revealed that the rgg gene in strain MGAS315 (a serotype M3 strain associated with invasive disease) encodes a proline at amino acid position 103 (Rgg(103P)); in contrast, all other strains encode a serine at this position (Rgg(103S)). A caseinolytic assay and Western blotting indicated that strain MGAS315 does not produce SpeB in vitro. Gene-swapping experiments showed that the rgg gene of MGAS315 is solely responsible for the lack of SpeB expression. In contrast to Rgg(103S), Rgg(103P) does not bind to the speB promoter in gel shift assays, which correlates with a lack of speB expression. Despite its inability to activate speB expression, Rgg(103P) retains the ability to bind to DNA upstream of norA and to influence its expression. Overall, this study illustrates how variation at the rgg locus may contribute to the phenotypic diversity of S. pyogenes.
Figures

Similar articles
-
Growth phase-dependent modulation of Rgg binding specificity in Streptococcus pyogenes.J Bacteriol. 2012 Aug;194(15):3961-71. doi: 10.1128/JB.06709-11. Epub 2012 May 25. J Bacteriol. 2012. PMID: 22636768 Free PMC article.
-
Phosphorylation at the D53 but Not the T65 Residue of CovR Determines the Repression of rgg and speB Transcription in emm1- and emm49-Type Group A Streptococci.J Bacteriol. 2019 Jan 28;201(4):e00681-18. doi: 10.1128/JB.00681-18. Print 2019 Feb 15. J Bacteriol. 2019. PMID: 30478086 Free PMC article.
-
The rgg gene of Streptococcus pyogenes NZ131 positively influences extracellular SPE B production.Infect Immun. 1999 Apr;67(4):1715-22. doi: 10.1128/IAI.67.4.1715-1722.1999. Infect Immun. 1999. PMID: 10085009 Free PMC article.
-
From transcription to activation: how group A streptococcus, the flesh-eating pathogen, regulates SpeB cysteine protease production.Mol Microbiol. 2011 Aug;81(3):588-601. doi: 10.1111/j.1365-2958.2011.07709.x. Epub 2011 Jun 24. Mol Microbiol. 2011. PMID: 21707787 Review.
-
Cysteine proteinase SpeB from Streptococcus pyogenes - a potent modifier of immunologically important host and bacterial proteins.Biol Chem. 2011 Dec;392(12):1077-88. doi: 10.1515/BC.2011.208. Biol Chem. 2011. PMID: 22050223 Review.
Cited by
-
Mutations in the control of virulence sensor gene from Streptococcus pyogenes after infection in mice lead to clonal bacterial variants with altered gene regulatory activity and virulence.PLoS One. 2014 Jun 26;9(6):e100698. doi: 10.1371/journal.pone.0100698. eCollection 2014. PLoS One. 2014. PMID: 24968349 Free PMC article.
-
The Mga Regulon but Not Deoxyribonuclease Sda1 of Invasive M1T1 Group A Streptococcus Contributes to In Vivo Selection of CovRS Mutations and Resistance to Innate Immune Killing Mechanisms.Infect Immun. 2015 Nov;83(11):4293-303. doi: 10.1128/IAI.00857-15. Epub 2015 Aug 17. Infect Immun. 2015. PMID: 26283338 Free PMC article.
-
Quorum sensing in group A Streptococcus.Front Cell Infect Microbiol. 2014 Sep 12;4:127. doi: 10.3389/fcimb.2014.00127. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25309879 Free PMC article. Review.
-
The Streptococcus pyogenes proteome: maps, virulence factors and vaccine candidates.Future Microbiol. 2010 Oct;5(10):1539-51. doi: 10.2217/fmb.10.116. Future Microbiol. 2010. PMID: 21073313 Free PMC article. Review.
-
Serotype M3 and M28 Group A Streptococci Have Distinct Capacities to Evade Neutrophil and TNF-α Responses and to Invade Soft Tissues.PLoS One. 2015 Jun 5;10(6):e0129417. doi: 10.1371/journal.pone.0129417. eCollection 2015. PLoS One. 2015. PMID: 26047469 Free PMC article.
References
-
- Aziz, R. K., M. J. Pabst, A. Jeng, R. Kansal, D. E. Low, V. Nizet, and M. Kotb. 2004. Invasive M1T1 group A Streptococcus undergoes a phase-shift in vivo to prevent proteolytic degradation of multiple virulence factors by SpeB. Mol. Microbiol. 51:123-134. - PubMed
-
- Beres, S. B., G. L. Sylva, K. D. Barbian, B. Lei, J. S. Hoff, N. D. Mammarella, M. Y. Liu, J. C. Smoot, S. F. Porcella, L. D. Parkins, D. S. Campbell, T. M. Smith, J. K. McCormick, D. Y. Leung, P. M. Schlievert, and J. M. Musser. 2002. Genome sequence of a serotype M3 strain of group A Streptococcus: phage-encoded toxins, the high-virulence phenotype, and clone emergence. Proc. Natl. Acad. Sci. USA 99:10078-10083. - PMC - PubMed
-
- Bryan, E. M., T. Bae, M. Kleerebezem, and G. M. Dunny. 2000. Improved vectors for nisin-controlled expression in gram-positive bacteria. Plasmid 44:183-190. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

