The convergence of information about rewarding and aversive stimuli in single neurons
- PMID: 19759296
- PMCID: PMC2782596
- DOI: 10.1523/JNEUROSCI.1815-09.2009
The convergence of information about rewarding and aversive stimuli in single neurons
Abstract
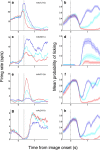
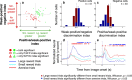
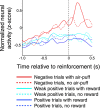
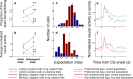
Neuroscientists, psychologists, clinicians, and economists have long been interested in how individuals weigh information about potential rewarding and aversive stimuli to make decisions and to regulate their emotions. However, we know relatively little about how appetitive and aversive systems interact in the brain, as most prior studies have investigated only one valence of reinforcement. Previous work has suggested that primate orbitofrontal cortex (OFC) represents information about the reward value of stimuli. We therefore investigated whether OFC also represents information about aversive stimuli, and, if so, whether individual neurons process information about both rewarding and aversive stimuli. Monkeys performed a trace conditioning task in which different novel abstract visual stimuli (conditioned stimuli, CSs) predicted the occurrence of one of three unconditioned stimuli (USs): a large liquid reward, a small liquid reward, or an aversive air-puff. Three lines of evidence suggest that information about rewarding and aversive stimuli converges in individual neurons in OFC. First, OFC neurons often responded to both rewarding and aversive USs, despite their different sensory features. Second, OFC neural responses to CSs often encoded information about both potential rewarding and aversive stimuli, even though these stimuli differed in both valence and sensory modality. Finally, OFC neural responses were correlated with monkeys' behavioral use of information about both rewarding and aversive CS-US associations. These data indicate that processing of appetitive and aversive stimuli converges at the single cell level in OFC, providing a possible substrate for executive and emotional processes that require using information from both appetitive and aversive systems.
Figures






References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
