Immunization with HSV-1 glycoprotein C prevents immune evasion from complement and enhances the efficacy of an HSV-1 glycoprotein D subunit vaccine
- PMID: 19761834
- PMCID: PMC2783579
- DOI: 10.1016/j.vaccine.2009.09.017
Immunization with HSV-1 glycoprotein C prevents immune evasion from complement and enhances the efficacy of an HSV-1 glycoprotein D subunit vaccine
Abstract
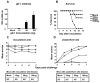
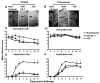
Herpes simplex virus type 1 (HSV-1) glycoprotein C (gC-1) binds complement component C3b and inhibits complement-mediated immunity. HSV-1 glycoprotein D (gD-1) is a potent immunogen and a candidate antigen for a subunit vaccine. We evaluated whether combined immunization with gD-1 and gC-1 provides better protection against challenge than gD-1 alone based on antibodies to gC-1 preventing HSV-1-mediated immune evasion. IgG purified from mice immunized with gC-1 blocked C3b binding to gC-1 and greatly increased neutralization by gD-1 IgG in the presence of complement. Passive transfer of gC-1 IgG protected complement intact mice against HSV-1 challenge but not C3 knockout mice, indicating that gC-1 antibody activity in vivo is complement-dependent. Immunizing mice with gD-1 and gC-1 provided better protection than gD-1 alone in preventing zosteriform disease and infection of dorsal root ganglia. Therefore, gC-1 immunization prevents HSV-1 evasion from complement and enhances the protection provided by gD-1 immunization.
Figures
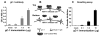





References
-
- Carroll MC. The role of complement in B cell activation and tolerance. Advances in Immunology. 2000;74:61–88. - PubMed
-
- Fang C, Miwa T, Shen H, Song WC. Complement-dependent enhancement of CD8+ T cell immunity to lymphocytic choriomeningitis virus infection in decay-accelerating factor-deficient mice. J Immunol. 2007;179(5):3178–86. erratum appears in J Immunol. 2007 Nov 15;179(10):7185. - PubMed
-
- Kopf M, Abel B, Gallimore A, Carroll M, Bachmann MF. Complement component C3 promotes T-cell priming and lung migration to control acute influenza virus infection. Nature Medicine. 2002;8(4):373–8. - PubMed
-
- Lachmann PJ, Davies A. Complement and immunity to viruses. Immunological Reviews. 1997;159:69–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

