Emerging concepts in acute mountain sickness and high-altitude cerebral edema: from the molecular to the morphological
- PMID: 19763397
- PMCID: PMC3085779
- DOI: 10.1007/s00018-009-0145-9
Emerging concepts in acute mountain sickness and high-altitude cerebral edema: from the molecular to the morphological
Abstract
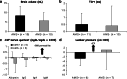
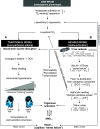
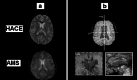
Acute mountain sickness (AMS) is a neurological disorder that typically affects mountaineers who ascend to high altitude. The symptoms have traditionally been ascribed to intracranial hypertension caused by extracellular vasogenic edematous brain swelling subsequent to mechanical disruption of the blood-brain barrier in hypoxia. However, recent diffusion-weighted magnetic resonance imaging studies have identified mild astrocytic swelling caused by a net redistribution of fluid from the "hypoxia-primed" extracellular space to the intracellular space without any evidence for further barrier disruption or additional increment in brain edema, swelling or pressure. These findings and the observation of minor vasogenic edema present in individuals with and without AMS suggest that the symptoms are not explained by cerebral edema. This has led to a re-evaluation of the relevant pathogenic events with a specific focus on free radicals and their interaction with the trigeminovascular system.
Figures
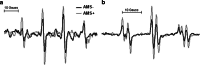
References
-
- Roach RC, Hackett PH. Frontiers of hypoxia research: acute mountain sickness. J Exp Biol. 2001;204:3161–3170. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

