Killing me softly: chlamydial use of proteolysis for evading host defenses
- PMID: 19765998
- PMCID: PMC2755597
- DOI: 10.1016/j.tim.2009.07.007
Killing me softly: chlamydial use of proteolysis for evading host defenses
Abstract
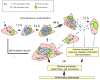
Chlamydial infections in humans cause severe health problems, including blinding trachoma and sexually transmitted diseases. Although the involved pathogenic mechanisms remain unclear, the ability to replicate and maintain long-term residence in the infected cells seems to significantly contribute to chlamydial pathogenicity. These obligate intracellular parasites maintain a delicate balance between exploiting and protecting their host: they occupy intracellular space and acquire nutrients from the infected cells, but at the same time they have to maintain the integrity of the host cells for the completion of their intracellular growth. For this purpose, chlamydiae hijack certain signaling pathways that prevent the host cells from undergoing apoptosis induced by intracellular stress and protect the infected cells from recognition and attack by host defenses. Interestingly, one of the strategies that chlamydiae use for these purposes is the induction of limited proteolysis of host proteins, which is the main focus of this article.
Figures
Similar articles
-
Chlamydia cell biology and pathogenesis.Nat Rev Microbiol. 2016 Jun;14(6):385-400. doi: 10.1038/nrmicro.2016.30. Epub 2016 Apr 25. Nat Rev Microbiol. 2016. PMID: 27108705 Free PMC article. Review.
-
New insights into Chlamydiae persistence: an energy metabolism strategy?Int J Immunopathol Pharmacol. 2013 Apr-Jun;26(2):525-8. doi: 10.1177/039463201302600227. Int J Immunopathol Pharmacol. 2013. PMID: 23755769
-
Chlamydial intracellular survival strategies.Cold Spring Harb Perspect Med. 2013 May 1;3(5):a010256. doi: 10.1101/cshperspect.a010256. Cold Spring Harb Perspect Med. 2013. PMID: 23637308 Free PMC article. Review.
-
Subversion of Cell-Autonomous Host Defense by Chlamydia Infection.Curr Top Microbiol Immunol. 2018;412:81-106. doi: 10.1007/82_2016_13. Curr Top Microbiol Immunol. 2018. PMID: 27169422 Review.
-
Effector protein modulation of host cells: examples in the Chlamydia spp. arsenal.Curr Opin Microbiol. 2009 Feb;12(1):81-7. doi: 10.1016/j.mib.2008.11.009. Epub 2009 Jan 8. Curr Opin Microbiol. 2009. PMID: 19138553 Review.
Cited by
-
Analysis of CPAF mutants: new functions, new questions (the ins and outs of a chlamydial protease).Pathog Dis. 2014 Aug;71(3):287-91. doi: 10.1111/2049-632X.12194. Pathog Dis. 2014. PMID: 24942261 Free PMC article.
-
Reassessing the role of the secreted protease CPAF in Chlamydia trachomatis infection through genetic approaches.Pathog Dis. 2014 Aug;71(3):336-51. doi: 10.1111/2049-632X.12179. Epub 2014 May 16. Pathog Dis. 2014. PMID: 24838663 Free PMC article.
-
Chlamydial protease-like activity factor targets SLC7A11 for degradation to induce ferroptosis and facilitate progeny releases.PLoS Pathog. 2025 Apr 8;21(4):e1013060. doi: 10.1371/journal.ppat.1013060. eCollection 2025 Apr. PLoS Pathog. 2025. PMID: 40198689 Free PMC article.
-
Immunity and vaccines against sexually transmitted Chlamydia trachomatis infection.Curr Opin Infect Dis. 2011 Feb;24(1):56-61. doi: 10.1097/QCO.0b013e3283421081. Curr Opin Infect Dis. 2011. PMID: 21124214 Free PMC article. Review.
-
Induction and inhibition of CPAF activity during analysis of Chlamydia-infected cells.Pathog Dis. 2015 Feb;73(1):1-8. doi: 10.1093/femspd/ftv007. Epub 2015 Feb 6. Pathog Dis. 2015. PMID: 25663342 Free PMC article.
References
-
- Everett KD, et al. Emended description of the order Chlamydiales, proposal of Parachlamydiaceae fam. nov. and Simkaniaceae fam. nov., each containing one monotypic genus, revised taxonomy of the family Chlamydiaceae, including a new genus and five new species, and standards for the identification of organisms. Int J Syst Bacteriol. 1999;49(Pt 2):415–440. - PubMed
-
- Stephens RS, et al. Genome sequence of an obligate intracellular pathogen of humans: Chlamydia trachomatis. Science. 1998;282:754–759. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

