Abeta42-to-Abeta40- and angiotensin-converting activities in different domains of angiotensin-converting enzyme
- PMID: 19773553
- PMCID: PMC2797262
- DOI: 10.1074/jbc.M109.011437
Abeta42-to-Abeta40- and angiotensin-converting activities in different domains of angiotensin-converting enzyme
Abstract
Amyloid beta-protein 1-42 (Abeta42) is believed to play a causative role in the development of Alzheimer disease (AD), although it is a minor part of Abeta. In contrast, Abeta40 is the predominant secreted form of Abeta and recent studies have suggested that Abeta40 has neuroprotective effects and inhibits amyloid deposition. We have reported that angiotensin-converting enzyme (ACE) converts Abeta42 to Abeta40, and its inhibition enhances brain Abeta42 deposition (Zou, K., Yamaguchi, H., Akatsu, H., Sakamoto, T., Ko, M., Mizoguchi, K., Gong, J. S., Yu, W., Yamamoto, T., Kosaka, K., Yanagisawa, K., and Michikawa, M. (2007) J. Neurosci. 27, 8628-8635). ACE has two homologous domains, each having a functional active site. In the present study, we identified the domain of ACE, which is responsible for converting Abeta42 to Abeta40. Interestingly, Abeta42-to-Abeta40-converting activity is solely found in the N-domain of ACE and the angiotensin-converting activity is found predominantly in the C-domain of ACE. We also found that the N-linked glycosylation is essential for both Abeta42-to-Abeta40- and angiotensin-converting activities and that unglycosylated ACE rapidly degraded. The domain-specific converting activity of ACE suggests that ACE inhibitors could be designed to specifically target the angiotensin-converting C-domain, without inhibiting the Abeta42-to-Abeta40-converting activity of ACE or increasing neurotoxic Abeta42.
Figures



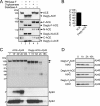
References
-
- Kehoe P. G., Wilcock G. K. (2007) Lancet Neurol. 6, 373–378 - PubMed
-
- Zou K., Michikawa M. (2008) Rev. Neurosci. 19, 203–212 - PubMed
-
- Lehmann D. J., Cortina-Borja M., Warden D. R., Smith A. D., Sleegers K., Prince J. A., van Duijn C. M., Kehoe P. G. (2005) Am. J. Epidemiol. 162, 305–317 - PubMed
-
- Kehoe P. G., Russ C., McIlory S., Williams H., Holmans P., Holmes C., Liolitsa D., Vahidassr D., Powell J., McGleenon B., Liddell M., Plomin R., Dynan K., Williams N., Neal J., Cairns N. J., Wilcock G., Passmore P., Lovestone S., Williams J., Owen M. J. (1999) Nat. Genet. 21, 71–72 - PubMed
-
- Elkins J. S., Douglas V. C., Johnston S. C. (2004) Neurology 62, 363–368 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

