Weekly administration of sagopilone (ZK-EPO), a fully synthetic epothilone, in patients with refractory solid tumours: results of a phase I trial
- PMID: 19773753
- PMCID: PMC2768435
- DOI: 10.1038/sj.bjc.6605327
Weekly administration of sagopilone (ZK-EPO), a fully synthetic epothilone, in patients with refractory solid tumours: results of a phase I trial
Abstract
Background: Epothilones are a novel class of microtubule-stabilising agents, and sagopilone is a fully synthetic epothilone that has shown marked in vivo and in vitro preclinical activity.
Methods: This phase I, open-label study investigated the maximum tolerated dose (MTD) and dose-limiting toxicities (DLTs) of weekly sagopilone. Twenty-three patients with malignancy resistant or refractory to standard treatment were enrolled into this study evaluating sagopilone doses from 0.6 to 7.0 mg m(-2).
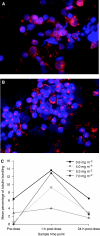
Results: The incidence of drug-related haematological adverse events (AEs) was low, with two grade 3 events observed. Nonhaematological AEs were generally mild and reversible; increased gamma-GT was the only grade 4 event and grade 3 events comprised peripheral neuropathy (n=2), diarrhoea (n=1) and fatigue (n=1). Two grade 3 events were DLTs (diarrhoea and peripheral neuropathy at 7.0 mg m(-2)). The MTD of weekly sagopilone was therefore established as 5.3 mg m(-2). Stable disease was the best overall response (n=3). Microtubule bundle formation in peripheral blood mononuclear cells increased post-treatment, peaking after 1 h. Sagopilone disposition was similar across treatment courses and showed rapidly decreasing serum concentrations after infusion end and a long terminal disposition phase with no obvious accumulation in the serum, probably reflecting a fast uptake into tissues followed by a slow release.
Conclusion: Weekly administration of sagopilone could represent an alternative to the 3-weekly administration currently evaluated in phase II trials.
Figures

References
-
- Goldspiel BR (1997) Clinical overview of the taxanes. Pharmacotherapy 17: 110S–125S - PubMed
-
- Graff J, Smith DC, Neerukonda L, Alonso M, Edgar A, Wang Y, Beer TM, The Prostate Cancer Clinical Trials Consortium (2008) Phase II study of sagopilone (ZK-EPO) plus prednisone as first-line chemotherapy in patients with metastatic androgen-independent prostate cancer 44th ASCO Annual Meeting, Chicago, Illinois, USA, May 30–June 3, Poster
-
- Hoffmann J, Vitale I, Buchmann B, Galluzzi L, Schwede W, Senovilla L, Skuballa W, Vivet S, Lichtner RB, Vicencio JM, Panaretakis T, Siemeister G, Lage H, Nanty L, Hammer S, Mittelstaedt K, Winsel S, Eschenbrenner J, Castedo M, Demarche C, Klar U, Kroemer G (2008) Improved cellular pharmacokinetics and pharmacodynamics underlie the wide anticancer activity of sagopilone. Cancer Res 68: 5301–5308 - PubMed
-
- Horwitz SB, Cohen D, Rao S, Ringel I, Shen H-J, Yang C-PH (1993) Taxol: mechanisms of action and resistance. J Natl Cancer Inst Monogr 15: 55–61 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

