Modulation of visual signals in macaque MT and MST neurons during pursuit eye movement
- PMID: 19776359
- PMCID: PMC2804434
- DOI: 10.1152/jn.90692.2008
Modulation of visual signals in macaque MT and MST neurons during pursuit eye movement
Abstract
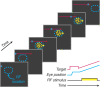
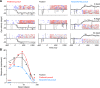
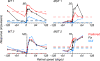
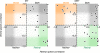
Retinal image motion is produced with each eye movement, yet we usually do not perceive this self-produced "reafferent" motion, nor are motion judgments much impaired when the eyes move. To understand the neural mechanisms involved in processing reafferent motion and distinguishing it from the motion of objects in the world, we studied the visual responses of single cells in middle temporal (MT) and medial superior temporal (MST) areas during steady fixation and smooth-pursuit eye movements in awake, behaving macaques. We measured neuronal responses to random-dot patterns moving at different speeds in a stimulus window that moved with the pursuit target and the eyes. This allowed us to control retinal image motion at all eye velocities. We found the expected high proportion of cells selective for the direction of visual motion. Pursuit tracking changed both response amplitude and preferred retinal speed for some cells. The changes in preferred speed were on average weakly but systematically related to the speed of pursuit for area MST cells, as would be expected if the shifts in speed selectivity were compensating for reafferent input. In area MT, speed tuning did not change systematically during pursuit. Many cells in both areas also changed response amplitude during pursuit; the most common form of modulation was response suppression when pursuit was opposite in direction to the cell's preferred direction. These results suggest that some cells in area MST encode retinal image motion veridically during eye movements, whereas others in both MT and MST contribute to the suppression of visual responses to reafferent motion.
Figures

References
-
- Albright TD. Direction and orientation selectivity of neurons in area MT of the macaque. J Neurophysiol 52: 1106–1129, 1984 - PubMed
-
- Albright TD. Centrifugal directional bias in the middle temporal visual area (MT) of the macaque. Vis Neurosci 2: 177–188, 1989 - PubMed
-
- Andersen RA, Essick GK, Siegel RM. Encoding of spatial location by posterior parietal neurons. Science 230: 456–458, 1985 - PubMed
-
- Andersen RA, Snyder LH, Li CS, Stricanne B. Coordinate transformations in the representation of spatial information. Curr Opin Neurobiol 3: 171–176, 1993 - PubMed
-
- Bair W, O'Keefe LP. The influence of fixational eye movements in the response of neurons in area MT of the macaque. Vis Neurosci 15: 779–186, 1998 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

