Morphogenesis of epithelial tubes: Insights into tube formation, elongation, and elaboration
- PMID: 19778532
- PMCID: PMC2854166
- DOI: 10.1016/j.ydbio.2009.09.024
Morphogenesis of epithelial tubes: Insights into tube formation, elongation, and elaboration
Abstract
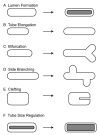
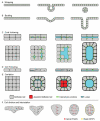
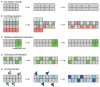
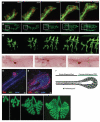
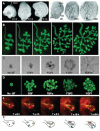
Epithelial tubes are a fundamental tissue across the metazoan phyla and provide an essential functional component of many of the major organs. Recent work in flies and mammals has begun to elucidate the cellular mechanisms driving the formation, elongation, and branching morphogenesis of epithelial tubes during development. Both forward and reverse genetic techniques have begun to identify critical molecular regulators for these processes and have revealed the conserved role of key pathways in regulating the growth and elaboration of tubular networks. In this review, we discuss the developmental programs driving the formation of branched epithelial networks, with specific emphasis on the trachea and salivary gland of Drosophila melanogaster and the mammalian lung, mammary gland, kidney, and salivary gland. We both highlight similarities in the development of these organs and attempt to identify tissue and organism specific strategies. Finally, we briefly consider how our understanding of the regulation of proliferation, apicobasal polarity, and epithelial motility during branching morphogenesis can be applied to understand the pathologic dysregulation of these same processes during metastatic cancer progression.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures

References
-
- Abrams EW, Mihoulides WK, Andrew DJ. Fork head and Sage maintain a uniform and patent salivary gland lumen through regulation of two downstream target genes, PH4alphaSG1 and PH4alphaSG2. Development. 2006;133:3517–27. - PubMed
-
- Adler PN. Planar signaling and morphogenesis in Drosophila. Dev Cell. 2002;2:525–35. - PubMed
-
- Araujo SJ, Aslam H, Tear G, Casanova J. mummy/cystic encodes an enzyme required for chitin and glycan synthesis, involved in trachea, embryonic cuticle and CNS development--analysis of its role in Drosophila tracheal morphogenesis. Dev Biol. 2005;288:179–93. - PubMed
-
- Bagnat M, Cheung ID, Mostov KE, Stainier DY. Genetic control of single lumen formation in the zebrafish gut. Nat Cell Biol. 2007;9:954–60. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

