RelA/p65 functions to maintain cellular senescence by regulating genomic stability and DNA repair
- PMID: 19779484
- PMCID: PMC2775178
- DOI: 10.1038/embor.2009.197
RelA/p65 functions to maintain cellular senescence by regulating genomic stability and DNA repair
Abstract
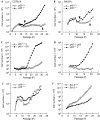
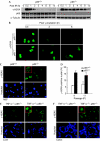
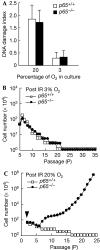
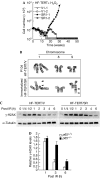
Nuclear factor (NF)-kappaB is a positive regulator of tumour development and progression, but how it functions in normal cells leading to oncogenesis is not clear. As cellular senescence has proven to be an intrinsic tumour suppressor mechanism that cells must overcome to establish deregulated growth, we used primary fibroblasts to follow NF-kappaB function in cells transitioning from senescence to subsequent immortalization. Our findings show that RelA/p65(-/-) murine fibroblasts immortalize at considerably faster rates than RelA/p65(+/+) cells. The ability of RelA/p65(-/-) fibroblasts to escape senescence earlier is due to their genomic instability, characterized by high frequencies of DNA mutations, gene deletions and gross chromosomal translocations. This increase in genomic instability is closely related to a compromised DNA repair that occurs in both murine RelA/p65(-/-) fibroblasts and tissues. Significantly, these results can also be duplicated in human fibroblasts lacking NF-kappaB. Altogether, our findings present a fresh perspective on the role of NF-kappaB as a tumour suppressor, which acts in pre-neoplastic cells to maintain cellular senescence by promoting DNA repair and genomic stability.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

References
-
- Bernard D, Gosselin K, Monte D, Vercamer C, Bouali F, Pourtier A, Vandenbunder B, Abbadie C (2004) Involvement of Rel/nuclear factor-kappaB transcription factors in keratinocyte senescence. Cancer Res 64: 472–481 - PubMed
-
- Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA (2005) Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436: 660–665 - PubMed
-
- Campisi J, d'Adda di Fagagna F (2007) Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 8: 729–740 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

