Spontaneous control of primary hepatitis C virus infection and immunity against persistent reinfection
- PMID: 19782080
- PMCID: PMC2889495
- DOI: 10.1053/j.gastro.2009.09.017
Spontaneous control of primary hepatitis C virus infection and immunity against persistent reinfection
Abstract
Background & aims: We followed patients with ongoing hepatitis C virus (HCV) exposure following control of an initial HCV infection to determine whether primary control conferred protection against future persistent infections.
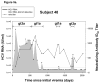
Methods: Twenty-two active injection drug users (IDU) who had cleared a primary hepatitis C viremia for at least 60 days were monitored monthly. Reinfection was defined as the detection of a new HCV infection. Protection was assessed based on the magnitude and duration of viremia following reinfection and generation of T-cell and neutralizing antibody (nAb) responses.
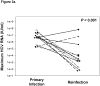
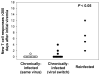
Results: Reinfection occurred in 11 IDU (50%) who previously spontaneously controlled primary HCV infection. Although viral clearance occurs in approximately 25% of patients with primary infections, spontaneous viral clearance was observed in 83% of reinfected patients. The duration and maximum level of viremia during subsequent episodes of reinfection were significantly decreased compared with those of the primary infection in the same subjects. In contrast to chronic infection, reinfection was associated with a significant increase in the breadth of T-cell responses. During acute infection, nAbs against heterologous viral pseudoparticles were detected in 60% of reinfected subjects; cross-reactive nAbs are rarely detected in patients who progress to chronic infection.
Conclusions: HCV reinfection is associated with a reduction in the magnitude and duration of viremia (compared with the initial infection), broadened cellular immune responses, and generation of cross-reactive humoral responses. These findings are consistent with development of adaptive immunity that is not sterilizing but protects against chronic disease.
Copyright 2010 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
There are no conflicts of interest to disclose.
Figures







References
-
- Cox A, Netski D, Mosbruger T, et al. Prospective evaluation of community-acquired acute-phase hepatitis C virus infection. Clin Infect Dis. 2005;40:951–8. - PubMed
-
- Prevention CfDCa. Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease. MMWR. 1998;47:1–39. - PubMed
-
- Garfein RS, Doherty MC, Monterroso ER, et al. Prevalence and incidence of hepatitis C virus infection among young adult injection drug users. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;18 (Suppl 1):S11–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

