Synthesis and biological evaluation of 14-(aminoalkyl-aminomethyl)aromathecins as topoisomerase I inhibitors: investigating the hypothesis of shared structure-activity relationships
- PMID: 19783447
- PMCID: PMC2769207
- DOI: 10.1016/j.bmc.2009.08.066
Synthesis and biological evaluation of 14-(aminoalkyl-aminomethyl)aromathecins as topoisomerase I inhibitors: investigating the hypothesis of shared structure-activity relationships
Abstract
The aromathecin topoisomerase I (top1) inhibitors offer promising scaffolds for the development of novel cancer chemotherapeutics. They are 'composites' of the camptothecin and indenoisoquinoline top1 inhibitors. Interestingly, some structure-activity relationship (SAR) overlap between the aromathecins and the indenoisoquinolines has been observed. For both classes, placement of certain polar groups in similar regions of the heteroaromatic system improves top1 inhibitory and antiproliferative activities. A series of water-soluble aromathecins substituted at position 14 with diaminoalkanes of various lengths has been prepared. These compounds all possess similar antiproliferative potency, but a general trend is observed: aromathecins with longer diaminoalkane substituents (>6 carbons) possess lower anti-top1 activity than their smaller counterparts (2-4 carbons), presumably as a result of unfavorable hydrophobic interactions. This trend is also noted with the indenoisoquinolines, revealing additional SAR overlap that supports the hypothesis that there is a 'universal' top1 inhibitor SAR.
Figures
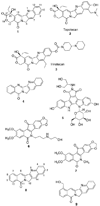
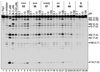


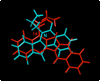

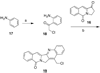
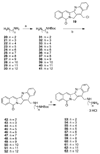
References
-
- Pommier Y. Nature Reviews Cancer. 2006;6:789. - PubMed
-
- Wang JC. Nature Rev. Mol. Cell Biol. 2002;3:430. - PubMed
-
- Husain I, Mohler JL, Seigler HF, Besterman JM. Cancer. Res. 1994;54(2):539. - PubMed
-
- Giovanella BC, Stehlin JS, Wall ME, Wani MC, Nicholas AW, Liu LF, Silber R, Potmesil M. Science. 1989;246:1046. - PubMed
-
- Wall ME, Wani MC, Cook CE, Palmer KH, A.T M, Sim GA. J. Am. Chem. Soc. 1966;88:3888. The.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

