Necl2 regulates epidermal adhesion and wound repair
- PMID: 19783739
- PMCID: PMC2752399
- DOI: 10.1242/dev.038232
Necl2 regulates epidermal adhesion and wound repair
Abstract
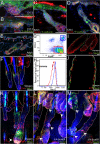
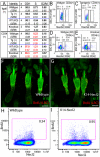
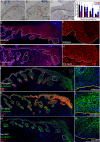
Differential expression of cell adhesion molecules regulates stem cell location, self-renewal and lineage selection under steady state conditions and during tissue repair. We show that the intercellular adhesion protein nectin-like molecule 2 (Necl2) is highly expressed in bulge stem cells of adult human and mouse hair follicles. Overexpression of Necl2 in cultured human keratinocytes led to upregulation of calcium/calmodulin-associated Ser/Thr kinase (CASK), increased calcium-independent intercellular adhesion, and inhibition of cell motility and in vitro wound healing. Although the rate of cell proliferation was reduced, terminal differentiation was unaffected. To assess the role of Necl2 in vivo, we examined the epidermis of Necl2-null mice and developed transgenic mice that expressed Necl2 in the basal layer of murine epidermis. Necl2 overexpression led to a reduction in S-phase cells and an increase in quiescent cells retaining DNA label in the bulge. Although epidermal homeostasis appeared normal in both transgenic and knockout mice, wound healing was markedly delayed. Necl2 overexpression resulted in reduced proliferation and increased levels of CASK and E-cadherin at the leading edge of healing wounds, consistent with its effects in culture. Our results demonstrate that Necl2 is involved in regulating epidermal stem cell quiescence and location.
Figures




References
-
- Bagutti, C., Hutter, C., Chiquet-Ehrismann, R., Fässler, R. and Watt, F. M. (2001). Dermal fibroblast-derived growth factors restore the ability of β1 integrin-deficient embryonal stem cells to differentiate into keratinocytes. Dev. Biol. 231, 321-333. - PubMed
-
- Benitah, S. A., Frye, M., Glogauer, M. and Watt, F. M. (2005). Stem cell depletion through epidermal deletion of Rac1. Science 309, 933-935. - PubMed
-
- Birchmeier, C., Birchmeier, W., Gherardi, E. and Vande Woude, G. F. (2003). Met, metastasis, motility and more. Nat. Rev. Mol. Cell Biol. 4, 915-925. - PubMed
-
- Blanpain, C., Lowry, W. E., Geoghegan, A., Polak, L. and Fuchs, E. (2004). Self-renewal, multipotency, and the existence of two cell populations within an epithelial stem cell niche. Cell 118, 635-648. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

