Total synthesis and antileishmanial activity of the natural occurring acetylenic fatty acids 6-heptadecynoic acid and 6-icosynoic acid
- PMID: 19789903
- PMCID: PMC2899701
- DOI: 10.1007/s11745-009-3345-z
Total synthesis and antileishmanial activity of the natural occurring acetylenic fatty acids 6-heptadecynoic acid and 6-icosynoic acid
Abstract
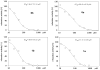
The first total syntheses of the naturally occurring acetylenic fatty acids-6-heptadecynoic acid (59% overall yield) and 6-icosynoic acid (34% overall yield)-was accomplished in four steps. Using the same synthetic sequence the naturally occurring fatty acids (6Z)-heptadecenoic acid (46% overall yield) and (6Z)-icosenoic acid (27% overall yield) were also synthesized. The Delta(6) acetylenic fatty acids displayed good antiprotozoal activity towards Leishmania donovani promastigotes (EC(50) = 1-6 microg/mL), but the 6-icosynoic acid was the most effective in the series. In addition, the (6Z)-icosenoic acid was a much better antiprotozoal compound (EC(50) = 5-6 microg/mL) than the (6Z)-heptadecenoic acid (EC(50) > 25 microg/mL). The saturated fatty acids n-heptadecanoic acid and n-eicosanoic acid were not effective towards L. donovani, indicating that the Delta(6) unsaturation in these fatty acids is necessary for leishmanicidal activity. In addition, both the 6-icosynoic acid and the (6Z)-icosenoic acid were inhibitors of the Leishmania DNA topoisomerase IB enzyme (EC(50's) = 36-49 microM), a possible intracellular target for these compounds. This is the first study assessing fatty acids as inhibitors of the Leishmania DNA topoisomerase IB enzyme.
Figures
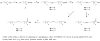
References
-
- Pearl MB, Kleiman R, Earle FR. Acetylenic acids of Alvaradoa amorphoides seed oil. Lipids. 1973;8:627–630. - PubMed
-
- Carballeira NM, Anastacio E, Salvá J, Ortega MJ. Identification of the new 10,15-eicosadienoic acid and related acids in the opisthobranch Haminaea templadoi. J Nat Prod. 1992;55:1783–1786. - PubMed
-
- Carballeira NM, Pagán M, Shalabi F, Nechev JT, Lahtchev K, Ivanova A, Stefanov K. Two novel iso-branched octadecenoic acids from a Micrococcus species. J Nat Prod. 2000;63:1573–1575. - PubMed
-
- Chaudhuri G, Ghoshal K, Banerjee AB. Toxic effects of fatty acids on Leishmania donovani promastigotes. Indian J Med Res. 1986;84:361–365. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

