Effect of calcium-modulating cyclophilin ligand on human immunodeficiency virus type 1 particle release and cell surface expression of tetherin
- PMID: 19793820
- PMCID: PMC2786830
- DOI: 10.1128/JVI.01786-09
Effect of calcium-modulating cyclophilin ligand on human immunodeficiency virus type 1 particle release and cell surface expression of tetherin
Abstract
The human immunodeficiency virus type 1 (HIV-1) accessory protein Vpu enhances virus particle release by counteracting a host factor that retains virions at the surfaces of infected cells. It was recently demonstrated that cellular protein BST-2/CD317/Tetherin restricts HIV-1 release in a Vpu-dependent manner. Calcium-modulating cyclophilin ligand (CAML) was also proposed to be involved in this process. We investigated whether CAML is involved in cell surface expression of Tetherin. Here, we show that CAML overexpression in permissive Cos-7 cells or CAML depletion in restrictive HeLa cells has no effect on HIV-1 release or on Tetherin surface expression, indicating that CAML is not required for Tetherin-mediated restriction of HIV-1 release.
Figures
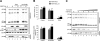
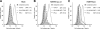
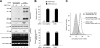

Similar articles
-
CAML does not modulate tetherin-mediated restriction of HIV-1 particle release.PLoS One. 2010 Feb 2;5(2):e9005. doi: 10.1371/journal.pone.0009005. PLoS One. 2010. PMID: 20126395 Free PMC article.
-
HIV-1 Vpu Antagonizes CD317/Tetherin by Adaptor Protein-1-Mediated Exclusion from Virus Assembly Sites.J Virol. 2016 Jul 11;90(15):6709-6723. doi: 10.1128/JVI.00504-16. Print 2016 Aug 1. J Virol. 2016. PMID: 27170757 Free PMC article.
-
Cell-cell spread of human immunodeficiency virus type 1 overcomes tetherin/BST-2-mediated restriction in T cells.J Virol. 2010 Dec;84(23):12185-99. doi: 10.1128/JVI.01447-10. Epub 2010 Sep 22. J Virol. 2010. PMID: 20861257 Free PMC article.
-
Tetherin/BST-2: Restriction Factor or Immunomodulator?Curr HIV Res. 2016;14(3):235-46. doi: 10.2174/1570162x14999160224102752. Curr HIV Res. 2016. PMID: 26957198 Free PMC article. Review.
-
Interactions of viral protein U (Vpu) with cellular factors.Curr Top Microbiol Immunol. 2009;339:27-45. doi: 10.1007/978-3-642-02175-6_2. Curr Top Microbiol Immunol. 2009. PMID: 20012522 Review.
Cited by
-
Assessing the Innate Sensing of HIV-1 Infected CD4+ T Cells by Plasmacytoid Dendritic Cells Using an Ex vivo Co-culture System.J Vis Exp. 2015 Sep 1;(103):51207. doi: 10.3791/51207. J Vis Exp. 2015. PMID: 26383704 Free PMC article.
-
CAML does not modulate tetherin-mediated restriction of HIV-1 particle release.PLoS One. 2010 Feb 2;5(2):e9005. doi: 10.1371/journal.pone.0009005. PLoS One. 2010. PMID: 20126395 Free PMC article.
-
Differential Control of BST2 Restriction and Plasmacytoid Dendritic Cell Antiviral Response by Antagonists Encoded by HIV-1 Group M and O Strains.J Virol. 2016 Oct 28;90(22):10236-10246. doi: 10.1128/JVI.01131-16. Print 2016 Nov 15. J Virol. 2016. PMID: 27581991 Free PMC article.
-
Virus-activated interferon regulatory factor 7 upregulates expression of the interferon-regulated BST2 gene independently of interferon signaling.J Virol. 2012 Apr;86(7):3513-27. doi: 10.1128/JVI.06971-11. Epub 2012 Feb 1. J Virol. 2012. PMID: 22301143 Free PMC article.
-
Unravelling the Immunomodulatory Effects of Viral Ion Channels, towards the Treatment of Disease.Viruses. 2021 Oct 27;13(11):2165. doi: 10.3390/v13112165. Viruses. 2021. PMID: 34834972 Free PMC article. Review.
References
-
- Bram, R. J., and G. R. Crabtree. 1994. Calcium signalling in T cells stimulated by a cyclophilin B-binding protein. Nature 371:355-358. - PubMed
-
- Cohen, E. A., E. F. Terwilliger, J. G. Sodroski, and W. A. Haseltine. 1988. Identification of a protein encoded by the vpu gene of HIV-1. Nature 334:532-534. - PubMed
-
- Cron, R. Q., S. R. Bartz, A. Clausell, S. J. Bort, S. J. Klebanoff, and D. B. Lewis. 2000. NFAT1 enhances HIV-1 gene expression in primary human CD4 T cells. Clin. Immunol. 94:179-191. - PubMed
-
- Dube, M., B. B. Roy, P. Guiot-Guillain, J. Mercier, J. Binette, G. Leung, and E. A. Cohen. 2009. Suppression of Tetherin-restricting activity upon human immunodeficiency virus type 1 particle release correlates with localization of Vpu in the trans-Golgi network. J. Virol. 83:4574-4590. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

