The African swine fever virus virion membrane protein pE248R is required for virus infectivity and an early postentry event
- PMID: 19793823
- PMCID: PMC2786719
- DOI: 10.1128/JVI.01333-09
The African swine fever virus virion membrane protein pE248R is required for virus infectivity and an early postentry event
Abstract
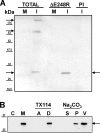
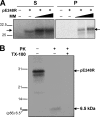
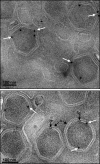
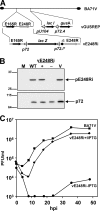
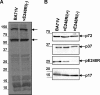
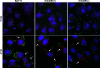
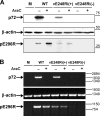
The African swine fever virus (ASFV) protein pE248R, encoded by the gene E248R, is a late structural component of the virus particle. The protein contains intramolecular disulfide bonds and has been previously identified as a substrate of the ASFV-encoded redox system. Its amino acid sequence contains a putative myristoylation site and a hydrophobic transmembrane region near its carboxy terminus. We show here that the protein pE248R is myristoylated during infection and associates with the membrane fraction in infected cells, behaving as an integral membrane protein. Furthermore, the protein localizes at the inner envelope of the virus particles in the cytoplasmic factories. The function of the protein pE248R in ASFV replication was investigated by using a recombinant virus that inducibly expresses the gene E248R. Under repressive conditions, the ASFV polyproteins pp220 and pp62 are normally processed and virus particles with morphology indistinguishable from that of those produced in a wild-type infection or under permissive conditions are generated. Moreover, the mutant virus particles can exit the cell as does the parental virus. However, the infectivity of the pE248R-deficient virions was reduced at least 100-fold. An investigation of the defect of the mutant virus indicated that neither virus binding nor internalization was affected by the absence of the protein pE248R, but a cytopathic effect was not induced and early and late gene expression was impaired, indicating that the protein is required for some early postentry event.
Figures




Similar articles
-
African Swine Fever Virus Protein pE199L Mediates Virus Entry by Enabling Membrane Fusion and Core Penetration.mBio. 2020 Aug 11;11(4):e00789-20. doi: 10.1128/mBio.00789-20. mBio. 2020. PMID: 32788374 Free PMC article.
-
African swine fever virus structural protein pE120R is essential for virus transport from assembly sites to plasma membrane but not for infectivity.J Virol. 2001 Aug;75(15):6758-68. doi: 10.1128/JVI.75.15.6758-6768.2001. J Virol. 2001. PMID: 11435554 Free PMC article.
-
African swine fever virus structural protein p54 is essential for the recruitment of envelope precursors to assembly sites.J Virol. 2004 Apr;78(8):4299-1313. doi: 10.1128/jvi.78.8.4299-4313.2004. J Virol. 2004. PMID: 15047843 Free PMC article.
-
African swine fever virus morphogenesis.Virus Res. 2013 Apr;173(1):29-41. doi: 10.1016/j.virusres.2012.09.016. Epub 2012 Oct 8. Virus Res. 2013. PMID: 23059353 Review.
-
African swine fever virus-cell interactions: from virus entry to cell survival.Virus Res. 2013 Apr;173(1):42-57. doi: 10.1016/j.virusres.2012.12.006. Epub 2012 Dec 20. Virus Res. 2013. PMID: 23262167 Free PMC article. Review.
Cited by
-
African Swine Fever Virus I267L Is a Hemorrhage-Related Gene Based on Transcriptome Analysis.Microorganisms. 2024 Feb 17;12(2):400. doi: 10.3390/microorganisms12020400. Microorganisms. 2024. PMID: 38399804 Free PMC article.
-
Spatiotemporally Orchestrated Interactions between Viral and Cellular Proteins Involved in the Entry of African Swine Fever Virus.Viruses. 2021 Dec 13;13(12):2495. doi: 10.3390/v13122495. Viruses. 2021. PMID: 34960765 Free PMC article. Review.
-
Deletion of the H240R Gene of African Swine Fever Virus Decreases Infectious Progeny Virus Production Due to Aberrant Virion Morphogenesis and Enhances Inflammatory Cytokine Expression in Porcine Macrophages.J Virol. 2022 Feb 9;96(3):e0166721. doi: 10.1128/JVI.01667-21. Epub 2021 Nov 17. J Virol. 2022. PMID: 34787458 Free PMC article.
-
Advances in African swine fever virus molecular biology and host interactions contributing to new tools for control.J Virol. 2025 Jun 17;99(6):e0093224. doi: 10.1128/jvi.00932-24. Epub 2025 May 9. J Virol. 2025. PMID: 40340396 Free PMC article. Review.
-
The Potential of Disabled Infectious Single Cycle (DISC) Virus Platforms for Next Generation African Swine Fever Vaccine Development.Transbound Emerg Dis. 2025 Jul 14;2025:8573171. doi: 10.1155/tbed/8573171. eCollection 2025. Transbound Emerg Dis. 2025. PMID: 40692871 Free PMC article. Review.
References
-
- Aguado, B., E. Viñuela, and A. Alcamí. 1991. African swine fever virus fatty acid acylated proteins. Virology 185:942-945. - PubMed
-
- Alcamí, A., A. L. Carrascosa, and E. Viñuela. 1989. Saturable binding sites mediate the entry of African swine fever virus into Vero cells. Virology 168:393-398. - PubMed
-
- Alcamí, A., A. L. Carrascosa, and E. Viñuela. 1989. The entry of African swine fever virus into Vero cells. Virology 171:68-75. - PubMed
-
- Alcamí, A., A. L. Carrascosa, and E. Viñuela. 1990. Interaction of African swine fever virus with macrophages. Virus Res. 17:93-104. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

