The ABC transporter PXA1 and peroxisomal beta-oxidation are vital for metabolism in mature leaves of Arabidopsis during extended darkness
- PMID: 19794119
- PMCID: PMC2768912
- DOI: 10.1105/tpc.108.064857
The ABC transporter PXA1 and peroxisomal beta-oxidation are vital for metabolism in mature leaves of Arabidopsis during extended darkness
Abstract
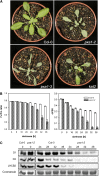
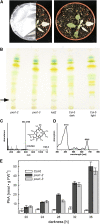
Fatty acid beta-oxidation is essential for seedling establishment of oilseed plants, but little is known about its role in leaf metabolism of adult plants. Arabidopsis thaliana plants with loss-of-function mutations in the peroxisomal ABC-transporter1 (PXA1) or the core beta-oxidation enzyme keto-acyl-thiolase 2 (KAT2) have impaired peroxisomal beta-oxidation. pxa1 and kat2 plants developed severe leaf necrosis, bleached rapidly when returned to light, and died after extended dark treatment, whereas the wild type was unaffected. Dark-treated pxa1 plants showed a decrease in photosystem II efficiency early on and accumulation of free fatty acids, mostly alpha-linolenic acid [18:3(n-3)] and pheophorbide a, a phototoxic chlorophyll catabolite causing the rapid bleaching. Isolated wild-type and pxa1 chloroplasts challenged with comparable alpha-linolenic acid concentrations both showed an 80% reduction in photosynthetic electron transport, whereas intact pxa1 plants were more susceptible to the toxic effects of alpha-linolenic acid than the wild type. Furthermore, starch-free mutants with impaired PXA1 function showed the phenotype more quickly, indicating a link between energy metabolism and beta-oxidation. We conclude that the accumulation of free polyunsaturated fatty acids causes membrane damage in pxa1 and kat2 plants and propose a model in which fatty acid respiration via peroxisomal beta-oxidation plays a major role in dark-treated plants after depletion of starch reserves.
Figures






Comment in
-
Nocturnal energy demand in plants: insights from studying mutants impaired in β-oxidation.Plant Signal Behav. 2010 Jul;5(7):842-4. doi: 10.4161/psb.5.7.11821. Epub 2010 Jul 1. Plant Signal Behav. 2010. PMID: 20505361 Free PMC article.
Similar articles
-
The α/β hydrolase CGI-58 and peroxisomal transport protein PXA1 coregulate lipid homeostasis and signaling in Arabidopsis.Plant Cell. 2013 May;25(5):1726-39. doi: 10.1105/tpc.113.111898. Epub 2013 May 10. Plant Cell. 2013. PMID: 23667126 Free PMC article.
-
The Arabidopsis pxa1 mutant is defective in an ATP-binding cassette transporter-like protein required for peroxisomal fatty acid beta-oxidation.Plant Physiol. 2001 Nov;127(3):1266-78. Plant Physiol. 2001. PMID: 11706205 Free PMC article.
-
Peroxisomal Acyl-CoA synthetase activity is essential for seedling development in Arabidopsis thaliana.Plant Cell. 2004 Feb;16(2):394-405. doi: 10.1105/tpc.019646. Epub 2004 Jan 23. Plant Cell. 2004. PMID: 14742880 Free PMC article.
-
Peroxisomal ABC transporters: functions and mechanism.Biochem Soc Trans. 2015 Oct;43(5):959-65. doi: 10.1042/BST20150127. Epub 2015 Oct 9. Biochem Soc Trans. 2015. PMID: 26517910 Free PMC article. Review.
-
Peroxisomal ABC transporters.FEBS Lett. 2006 Feb 13;580(4):1139-55. doi: 10.1016/j.febslet.2005.12.095. Epub 2006 Jan 9. FEBS Lett. 2006. PMID: 16413537 Review.
Cited by
-
Arabidopsis peroxisome proteomics.Front Plant Sci. 2013 Apr 24;4:101. doi: 10.3389/fpls.2013.00101. eCollection 2013. Front Plant Sci. 2013. PMID: 23630535 Free PMC article.
-
Quantitative analysis of organelle distribution and dynamics in Physcomitrella patens protonemal cells.BMC Plant Biol. 2012 May 17;12:70. doi: 10.1186/1471-2229-12-70. BMC Plant Biol. 2012. PMID: 22594499 Free PMC article.
-
The α/β hydrolase CGI-58 and peroxisomal transport protein PXA1 coregulate lipid homeostasis and signaling in Arabidopsis.Plant Cell. 2013 May;25(5):1726-39. doi: 10.1105/tpc.113.111898. Epub 2013 May 10. Plant Cell. 2013. PMID: 23667126 Free PMC article.
-
ABI4 activates DGAT1 expression in Arabidopsis seedlings during nitrogen deficiency.Plant Physiol. 2011 Jun;156(2):873-83. doi: 10.1104/pp.111.175950. Epub 2011 Apr 22. Plant Physiol. 2011. PMID: 21515696 Free PMC article.
-
A Biostimulant Obtained from the Seaweed Ascophyllum nodosum Protects Arabidopsis thaliana from Severe Oxidative Stress.Int J Mol Sci. 2020 Jan 11;21(2):474. doi: 10.3390/ijms21020474. Int J Mol Sci. 2020. PMID: 31940839 Free PMC article.
References
-
- Adham, A.R., Zolman, B.K., Millius, A., and Bartel, B. (2005). Mutations in Arabidopsis acyl-CoA oxidase genes reveal distinct and overlapping roles in beta-oxidation. Plant J. 41 859–874. - PubMed
-
- Alonso, J.M., et al. (2003). Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301 653–657. - PubMed
-
- Baker, A., Graham, I.A., Holdsworth, M., Smith, S.M., and Theodoulou, F.L. (2006). Chewing the fat: Beta-oxidation in signalling and development. Trends Plant Sci. 11 124–132. - PubMed
-
- Berger, J., and Gärtner, J. (2006). X-linked adrenoleukodystrophy: Clinical, biochemical and pathogenetic aspects. Biochim. Biophys. Acta 1763 1721–1732. - PubMed
-
- Bligh, E.G., and Dyer, W.J. (1959). A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37 911–917. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

