High-mobility group protein box-1 and its relevance to cerebral ischemia
- PMID: 19794402
- PMCID: PMC2949127
- DOI: 10.1038/jcbfm.2009.202
High-mobility group protein box-1 and its relevance to cerebral ischemia
Abstract
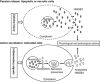
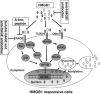
High-mobility group box-1 (HMGB1) was originally identified as a ubiquitously expressed, abundant, nonhistone DNA-binding protein. It has well-established functions in the maintenance of nuclear homeostasis. The HMGB1 can either be passively released into the extracellular milieu in response to necrotic signals or actively secreted in response to inflammatory signals. Extracellular HMGB1 interacts with receptors, including those for advanced glycation endproducts (RAGEs) as well as Toll-like receptor 2 (TLR2) and TLR4. The HMGB1 functions in a synergistic manner with other proinflammatory mediators and acts as a potent proinflammatory cytokine-like factor that contributes to the pathogenesis of diverse inflammatory and infectious disorders. Numerous reports point to HMGB1 as a novel player in the ischemic brain. This review provides an appraisal of the emerging roles of HMGB1 in cerebral ischemia injury, highlighting the relevance of HMGB1-blocking agents as potent therapeutic tools for neuroprotection.
Figures

References
-
- Agnello D, Wang H, Yang H, Tracey KJ, Ghezzi P. HMGB-1, a DNA-binding protein with cytokine activity, induces brain TNF and IL-6 production, and mediates anorexia and taste aversion. Cytokine. 2002;18:231–236. - PubMed
-
- Bianchi ME. Significant (re)location: how to use chromatin and/or abundant proteins as messages of life and death. Trends Cell Biol. 2004;14:287–293. - PubMed
-
- Bianchi ME, Manfredi AA. High-mobility group box 1 (HMGB1) protein at the crossroads between innate and adaptive immunity. Immunol Rev. 2007;220:35–46. - PubMed
-
- Bierhaus A, Humpert PM, Morcos M, Wendt T, Chavakis T, Arnold B, Stern DM, Nawroth PP. Understanding RAGE, the receptor for advanced glycation end products. J Mol Med. 2005;83:876–886. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

