Substrate interactions and promiscuity in a viral DNA packaging motor
- PMID: 19794496
- PMCID: PMC2769991
- DOI: 10.1038/nature08443
Substrate interactions and promiscuity in a viral DNA packaging motor
Abstract
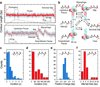
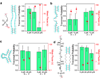
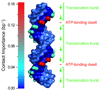
The ASCE (additional strand, conserved E) superfamily of proteins consists of structurally similar ATPases associated with diverse cellular activities involving metabolism and transport of proteins and nucleic acids in all forms of life. A subset of these enzymes consists of multimeric ringed pumps responsible for DNA transport in processes including genome packaging in adenoviruses, herpesviruses, poxviruses and tailed bacteriophages. Although their mechanism of mechanochemical conversion is beginning to be understood, little is known about how these motors engage their nucleic acid substrates. Questions remain as to whether the motors contact a single DNA element, such as a phosphate or a base, or whether contacts are distributed over several parts of the DNA. Furthermore, the role of these contacts in the mechanochemical cycle is unknown. Here we use the genome packaging motor of the Bacillus subtilis bacteriophage varphi29 (ref. 4) to address these questions. The full mechanochemical cycle of the motor, in which the ATPase is a pentameric-ring of gene product 16 (gp16), involves two phases-an ATP-loading dwell followed by a translocation burst of four 2.5-base-pair (bp) steps triggered by hydrolysis product release. By challenging the motor with a variety of modified DNA substrates, we show that during the dwell phase important contacts are made with adjacent phosphates every 10-bp on the 5'-3' strand in the direction of packaging. As well as providing stable, long-lived contacts, these phosphate interactions also regulate the chemical cycle. In contrast, during the burst phase, we find that DNA translocation is driven against large forces by extensive contacts, some of which are not specific to the chemical moieties of DNA. Such promiscuous, nonspecific contacts may reflect common translocase-substrate interactions for both the nucleic acid and protein translocases of the ASCE superfamily.
Figures

References
-
- Burroughs AM, Iyer LM, Aravind L. Basel: Karger; 2007. Comparative Genomics and Evolutionary Trajectories of Viral ATP Dependent DNA-Packaging Systems; pp. 48–65. - PubMed
-
- Hopfner KP, Michaelis J. Mechanisms of nucleic acid translocases: lessons from structural biology and single-molecule biophysics. Curr Opin Struct Biol. 2007;17:87–95. - PubMed
-
- Smith DE, et al. The bacteriophage phi29 portal motor can package DNA against a large internal force. Nature. 2001;413:748–752. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

