The Arabidopsis ATP-binding cassette protein AtMRP5/AtABCC5 is a high affinity inositol hexakisphosphate transporter involved in guard cell signaling and phytate storage
- PMID: 19797057
- PMCID: PMC2785203
- DOI: 10.1074/jbc.M109.030247
The Arabidopsis ATP-binding cassette protein AtMRP5/AtABCC5 is a high affinity inositol hexakisphosphate transporter involved in guard cell signaling and phytate storage
Abstract
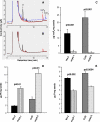
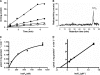
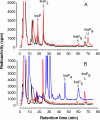
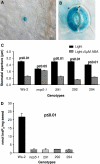
Arabidopsis possesses a superfamily of ATP-binding cassette (ABC) transporters. Among these, the multidrug resistance-associated protein AtMRP5/AtABCC5 regulates stomatal aperture and controls plasma membrane anion channels of guard cells. Remarkably, despite the prominent role of AtMRP5 in conferring partial drought insensitivity upon Arabidopsis, we know little of the biochemical function of AtMRP5. Our phylogenetic analysis showed that AtMRP5 is closely related to maize MRP4, mutation of which confers a low inositol hexakisphosphate kernel phenotype. We now show that insertion mutants of AtMRP5 display a low inositol hexakisphosphate phenotype in seed tissue and that this phenotype is associated with alterations of mineral cation and phosphate status. By heterologous expression in yeast, we demonstrate that AtMRP5 encodes a specific and high affinity ATP-dependent inositol hexakisphosphate transporter that is sensitive to inhibitors of ABC transporters. Moreover, complementation of the mrp5-1 insertion mutants of Arabidopsis with the AtMRP5 cDNA driven from a guard cell-specific promoter restores the sensitivity of the mutant to abscisic acid-mediated inhibition of stomatal opening. Additionally, we show that mutation of residues of the Walker B motif prevents restoring the multiple phenotypes associated with mrp5-1. Our findings highlight a novel function of plant ABC transporters that may be relevant to other kingdoms. They also extend the signaling repertoire of this ubiquitous inositol polyphosphate signaling molecule.
Figures
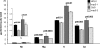

Similar articles
-
The ATP binding cassette transporter AtMRP5 modulates anion and calcium channel activities in Arabidopsis guard cells.J Biol Chem. 2007 Jan 19;282(3):1916-24. doi: 10.1074/jbc.M607926200. Epub 2006 Nov 10. J Biol Chem. 2007. PMID: 17098742
-
The plant multidrug resistance ABC transporter AtMRP5 is involved in guard cell hormonal signalling and water use.Plant J. 2003 Jan;33(1):119-29. doi: 10.1046/j.1365-313x.2003.016012.x. Plant J. 2003. PMID: 12943546
-
The Arabidopsis thaliana ABC transporter AtMRP5 controls root development and stomata movement.EMBO J. 2001 Apr 17;20(8):1875-87. doi: 10.1093/emboj/20.8.1875. EMBO J. 2001. PMID: 11296221 Free PMC article.
-
The guard cell as a single-cell model towards understanding drought tolerance and abscisic acid action.J Exp Bot. 2009;60(5):1439-63. doi: 10.1093/jxb/ern340. Epub 2009 Jan 30. J Exp Bot. 2009. PMID: 19181866 Review.
-
myo-Inositol-1,2,3,4,5,6-hexakisphosphate.Phytochemistry. 2003 Nov;64(6):1033-43. doi: 10.1016/s0031-9422(03)00446-1. Phytochemistry. 2003. PMID: 14568069 Review.
Cited by
-
The Arabidopsis thaliana MYB60 promoter provides a tool for the spatio-temporal control of gene expression in stomatal guard cells.J Exp Bot. 2013 Aug;64(11):3361-71. doi: 10.1093/jxb/ert180. Epub 2013 Jul 4. J Exp Bot. 2013. PMID: 23828545 Free PMC article.
-
MRP Transporters and Low Phytic Acid Mutants in Major Crops: Main Pleiotropic Effects and Future Perspectives.Front Plant Sci. 2020 Aug 27;11:1301. doi: 10.3389/fpls.2020.01301. eCollection 2020. Front Plant Sci. 2020. PMID: 32973854 Free PMC article. Review.
-
InsP6-sensitive variants of the Gle1 mRNA export factor rescue growth and fertility defects of the ipk1 low-phytic-acid mutation in Arabidopsis.Plant Cell. 2015 Feb;27(2):417-31. doi: 10.1105/tpc.114.132134. Epub 2015 Feb 10. Plant Cell. 2015. PMID: 25670768 Free PMC article.
-
Characterization of the Symbiotic Nitrogen-Fixing Common Bean Low Phytic Acid (lpa1) Mutant Response to Water Stress.Genes (Basel). 2018 Feb 15;9(2):99. doi: 10.3390/genes9020099. Genes (Basel). 2018. PMID: 29462877 Free PMC article.
-
Phytic acid in green leaves of herbaceous plants-temporal variation in situ and response to different nitrogen/phosphorus fertilizing regimes.AoB Plants. 2014 Aug 13;6:plu048. doi: 10.1093/aobpla/plu048. AoB Plants. 2014. PMID: 25125697 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

