Effect of the O-antigen length of lipopolysaccharide on the functions of Type III secretion systems in Salmonella enterica
- PMID: 19797066
- PMCID: PMC2786484
- DOI: 10.1128/IAI.00871-09
Effect of the O-antigen length of lipopolysaccharide on the functions of Type III secretion systems in Salmonella enterica
Abstract
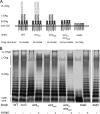
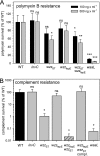
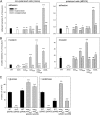
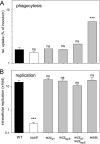
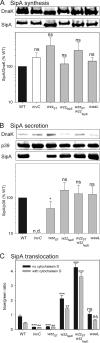
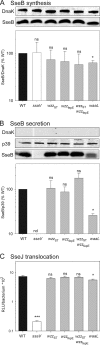
The virulence of Salmonella enterica critically depends on the functions of two type III secretion systems (T3SS), with the Salmonella pathogenicity island 1 (SPI1)-encoded T3SS required for host cell invasion and the SPI2-T3SS enabling Salmonella to proliferate within host cells. A further T3SS is required for the assembly of the flagella. Most serovars of Salmonella also possess a lipopolysaccharide with a complex O-antigen (OAg) structure. The number of OAg units attached to the core polysaccharide varies between 16 and more than 100 repeats, with a trimodal distribution. This work investigated the correlation of the OAg length with the functions of the SPI1-T3SS and the SPI2-T3SS. We observed that the number of repeats of OAg units had no effect on bacterial motility. The interaction of Salmonella with epithelial cells was altered if the OAg structure was changed by mutations in regulators of OAg. Strains defective in synthesis of very long or long and very long OAg species showed increased translocation of a SPI1-T3SS effector protein and increased invasion. Invasion of a strain entirely lacking OAg was increased, but this mutant strain also showed increased adhesion. In contrast, translocation of a SPI2-T3SS effector protein and intracellular replication were not affected by modification of the OAg length. Mutant strains lacking the entire OAg or long and very long OAg were highly susceptible to complement killing. These observations indicate that the architecture of the outer membrane of Salmonella is balanced to permit sufficient T3SS function but also to confer optimal protection against antimicrobial defense mechanisms.
Figures

Similar articles
-
Functional dissection of translocon proteins of the Salmonella pathogenicity island 2-encoded type III secretion system.BMC Microbiol. 2010 Apr 8;10:104. doi: 10.1186/1471-2180-10-104. BMC Microbiol. 2010. PMID: 20377892 Free PMC article.
-
Enoxacin adversely affects Salmonella enterica virulence and host pathogenesis through interference with type III secretion system type II (T3SS-II) and disruption of translocation of Salmonella Pathogenicity Island-2 (SPI2) effectors.J Microbiol. 2025 Feb;63(2):e2410015. doi: 10.71150/jm.2410015. Epub 2025 Feb 27. J Microbiol. 2025. PMID: 40044131
-
Novel Role of VisP and the Wzz System during O-Antigen Assembly in Salmonella enterica Serovar Typhimurium Pathogenesis.Infect Immun. 2018 Jul 23;86(8):e00319-18. doi: 10.1128/IAI.00319-18. Print 2018 Aug. Infect Immun. 2018. PMID: 29866904 Free PMC article.
-
Salmonella pathogenicity islands in host specificity, host pathogen-interactions and antibiotics resistance of Salmonella enterica.Berl Munch Tierarztl Wochenschr. 2007 Jul-Aug;120(7-8):317-27. Berl Munch Tierarztl Wochenschr. 2007. PMID: 17715824 Review.
-
Cellular microbiology of intracellular Salmonella enterica: functions of the type III secretion system encoded by Salmonella pathogenicity island 2.Cell Mol Life Sci. 2004 Nov;61(22):2812-26. doi: 10.1007/s00018-004-4248-z. Cell Mol Life Sci. 2004. PMID: 15558211 Free PMC article. Review.
Cited by
-
The small RNA STnc1480 contributes to the regulation of biofilm formation and pathogenicity in Salmonella typhimurium.Arch Microbiol. 2022 Nov 19;204(12):716. doi: 10.1007/s00203-022-03331-9. Arch Microbiol. 2022. PMID: 36400974
-
Association of a protective monoclonal IgA with the O antigen of Salmonella enterica serovar Typhimurium impacts type 3 secretion and outer membrane integrity.Infect Immun. 2012 Jul;80(7):2454-63. doi: 10.1128/IAI.00018-12. Epub 2012 Apr 2. Infect Immun. 2012. PMID: 22473607 Free PMC article.
-
The extradomain a of fibronectin enhances the efficacy of lipopolysaccharide defective Salmonella bacterins as vaccines in mice.Vet Res. 2012 Apr 19;43(1):31. doi: 10.1186/1297-9716-43-31. Vet Res. 2012. PMID: 22515195 Free PMC article.
-
Salmonella enterica Serovar Typhimurium Uses PbgA/YejM To Regulate Lipopolysaccharide Assembly during Bacteremia.Infect Immun. 2019 Dec 17;88(1):e00758-19. doi: 10.1128/IAI.00758-19. Print 2019 Dec 17. Infect Immun. 2019. PMID: 31611279 Free PMC article.
-
Bacterial outer membrane vesicles provide an alternative pathway for trafficking of Escherichia coli O157 type III secreted effectors to epithelial cells.mSphere. 2023 Dec 20;8(6):e0052023. doi: 10.1128/msphere.00520-23. Epub 2023 Nov 6. mSphere. 2023. PMID: 37929984 Free PMC article.
References
-
- Abeyrathne, P. D., and J. S. Lam. 2007. WaaL of Pseudomonas aeruginosa utilizes ATP in in vitro ligation of O antigen onto lipid A-core. Mol. Microbiol. 65:1345-1359. - PubMed
-
- Ackermann, M., B. Stecher, N. E. Freed, P. Songhet, W. D. Hardt, and M. Doebeli. 2008. Self-destructive cooperation mediated by phenotypic noise. Nature 454:987-990. - PubMed
-
- Ausubel, F. M., R. Brent, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, and K. Struhl. 1987. Current protocols in molecular biology. Wiley, New York, NY.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

