Promising bone-related therapeutic targets for rheumatoid arthritis
- PMID: 19798028
- PMCID: PMC2797318
- DOI: 10.1038/nrrheum.2009.175
Promising bone-related therapeutic targets for rheumatoid arthritis
Abstract
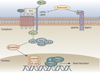
Rheumatoid arthritis (RA) is a chronic, debilitating autoimmune disease that results in inflammation and structural destruction of the joints. A hallmark of RA pathogenesis is an imbalance of the osteoblast-osteoclast axis driven by inflammatory processes, resulting in elevated bone resorption by osteoclasts. Current therapies used to treat this disease have focused on inhibition of synovitis, but such treatments do not adequately repair damaged bone. A key pathway of osteoclast formation involves the receptor activator of nuclear factor kappaB ligand (RANKL) pathway acting on myeloid progenitor cells. The Wnt pathway has been shown to be important for the differentiation of osteoblasts from mesenchymal lineage precursors, and endogenous Wnt inhibitors, such as Dickkopf1 and sclerostin, might have important roles in osteoclast dysregulation in RA. Inhibition of the RANKL pathway, or blockade of Dickkopf1 and sclerostin, might serve to restore the osteoblast-osteoclast balance and repair bone erosion in RA joints. Such treatments, in combination with anti-inflammatory therapies, could stabilize and repair damaged joints and have the potential to be valuable additions to the armory of RA treatments.
Figures

References
-
- Tak PP, Bresnihan B. The pathogenesis and prevention of joint damage in rheumatoid arthritis: advances from synovial biopsy and tissue analysis. Arthritis Rheum. 2000;43:2619–2633. - PubMed
-
- Walsh MC, et al. Osteoimmunology: interplay between the immune system and bone metabolism. Annu Rev Immunol. 2006;24:33–63. - PubMed
-
- Combe B. Progression in early rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2009;23:59–69. - PubMed
-
- Klareskog L, et al. Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial. Lancet. 2004;363:675–681. - PubMed
-
- Smolen JS, et al. Predictors of joint damage in patients with early rheumatoid arthritis treated with high-dose methotrexate with or without concomitant infliximab: results from the ASPIRE trial. Arthritis Rheum. 2006;54:702–710. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

