Interactome analyses identify ties of PrP and its mammalian paralogs to oligomannosidic N-glycans and endoplasmic reticulum-derived chaperones
- PMID: 19798432
- PMCID: PMC2749441
- DOI: 10.1371/journal.ppat.1000608
Interactome analyses identify ties of PrP and its mammalian paralogs to oligomannosidic N-glycans and endoplasmic reticulum-derived chaperones
Erratum in
- PLoS Pathog. 2009 Oct;5(10). doi: 10.1371/annotation/9eb11869-6acb-49b0-978e-abedc3cc545a. Won, Amy Hye [corrected to Jeon, Amy Hye Won]
Abstract
The physiological environment which hosts the conformational conversion of the cellular prion protein (PrP(C)) to disease-associated isoforms has remained enigmatic. A quantitative investigation of the PrP(C) interactome was conducted in a cell culture model permissive to prion replication. To facilitate recognition of relevant interactors, the study was extended to Doppel (Prnd) and Shadoo (Sprn), two mammalian PrP(C) paralogs. Interestingly, this work not only established a similar physiological environment for the three prion protein family members in neuroblastoma cells, but also suggested direct interactions amongst them. Furthermore, multiple interactions between PrP(C) and the neural cell adhesion molecule, the laminin receptor precursor, Na/K ATPases and protein disulfide isomerases (PDI) were confirmed, thereby reconciling previously separate findings. Subsequent validation experiments established that interactions of PrP(C) with PDIs may extend beyond the endoplasmic reticulum and may play a hitherto unrecognized role in the accumulation of PrP(Sc). A simple hypothesis is presented which accounts for the majority of interactions observed in uninfected cells and suggests that PrP(C) organizes its molecular environment on account of its ability to bind to adhesion molecules harboring immunoglobulin-like domains, which in turn recognize oligomannose-bearing membrane proteins.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



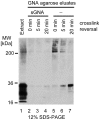
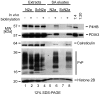

Similar articles
-
The CNS glycoprotein Shadoo has PrP(C)-like protective properties and displays reduced levels in prion infections.EMBO J. 2007 Sep 5;26(17):4038-50. doi: 10.1038/sj.emboj.7601830. Epub 2007 Aug 16. EMBO J. 2007. PMID: 17703189 Free PMC article.
-
Protein disulfide isomerase regulates endoplasmic reticulum stress and the apoptotic process during prion infection and PrP mutant-induced cytotoxicity.PLoS One. 2012;7(6):e38221. doi: 10.1371/journal.pone.0038221. Epub 2012 Jun 7. PLoS One. 2012. PMID: 22685557 Free PMC article.
-
Cellular prion protein is implicated in the regulation of local Ca2+ movements in cerebellar granule neurons.J Neurochem. 2011 Mar;116(5):881-90. doi: 10.1111/j.1471-4159.2010.07015.x. Epub 2011 Jan 7. J Neurochem. 2011. PMID: 21214552
-
The prion gene complex encoding PrP(C) and Doppel: insights from mutational analysis.Gene. 2001 Sep 5;275(1):1-18. doi: 10.1016/s0378-1119(01)00627-8. Gene. 2001. PMID: 11574147 Review.
-
The highways and byways of prion protein trafficking.Trends Cell Biol. 2005 Feb;15(2):102-11. doi: 10.1016/j.tcb.2004.12.002. Trends Cell Biol. 2005. PMID: 15695097 Review.
Cited by
-
Disulfide bonding in neurodegenerative misfolding diseases.Int J Cell Biol. 2013;2013:318319. doi: 10.1155/2013/318319. Epub 2013 Aug 1. Int J Cell Biol. 2013. PMID: 23983694 Free PMC article.
-
Protein disulphide isomerase (PDI) is protective against amyotrophic lateral sclerosis (ALS)-related mutant Fused in Sarcoma (FUS) in in vitro models.Sci Rep. 2021 Sep 2;11(1):17557. doi: 10.1038/s41598-021-96181-2. Sci Rep. 2021. PMID: 34475430 Free PMC article.
-
Recombinant human prion protein inhibits prion propagation in vitro.Sci Rep. 2013 Oct 9;3:2911. doi: 10.1038/srep02911. Sci Rep. 2013. PMID: 24105336 Free PMC article.
-
Knockout of the prion protein (PrP)-like Sprn gene does not produce embryonic lethality in combination with PrP(C)-deficiency.Proc Natl Acad Sci U S A. 2012 Jun 5;109(23):9035-40. doi: 10.1073/pnas.1202130109. Epub 2012 May 22. Proc Natl Acad Sci U S A. 2012. PMID: 22619325 Free PMC article.
-
Potential roles of the endoplasmic reticulum stress pathway in amyotrophic lateral sclerosis.Front Aging Neurosci. 2023 Feb 15;15:1047897. doi: 10.3389/fnagi.2023.1047897. eCollection 2023. Front Aging Neurosci. 2023. PMID: 36875699 Free PMC article. Review.
References
-
- Taylor DR, Hooper NM. The prion protein and lipid rafts. Mol Membr Biol. 2006;23:89–99. - PubMed
-
- Watts JC, Westaway D. The prion protein family: diversity, rivalry, and dysfunction. Biochim Biophys Acta. 2007;1772:654–672. - PubMed
-
- Ben-Zaken O, Tzaban S, Tal Y, Horonchik L, Esko JD, et al. Cellular heparan sulfate participates in the metabolism of prions. J Biol Chem. 2003;278:40041–40049. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

