Increase in tumor size following intratumoral injection of immunostimulatory CpG-containing oligonucleotides in a rat glioma model
- PMID: 19798500
- PMCID: PMC11029917
- DOI: 10.1007/s00262-009-0771-y
Increase in tumor size following intratumoral injection of immunostimulatory CpG-containing oligonucleotides in a rat glioma model
Abstract
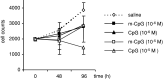
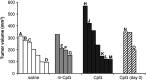
The immunosuppressive environment of malignant gliomas is likely to suppress the anti-tumor activity of infiltrating microglial cells and lymphocytes. Macrophages and microglial cells may be activated by oligonucleotides containing unmethylated CpG-motifs, although their value in cancer immunotherapy has remained controversial. Following injection of CpG-containing oligonucleotides (ODN) into normal rat brain, we observed a local inflammatory response with CD8+ T cell infiltration, upregulation of MHC 2, and ED1 expression proving the immunogenic capacity of the CpG-ODN used. This was not observed with a control ODN mutated in the immunostimulatory sequence (m-CpG). To study their effect in a syngeneic tumor model, we implanted rat 9L gliosarcoma cells into the striatum of Fisher 344 rats. After 3 days, immunostimulatory CpG-ODN, control m-CpG-ODN, or saline was injected stereotactically into the tumors (day 3 group). In another group of animals (day 0 group), CpG-ODN were mixed with 9L cells prior to implantation without further treatment on day 3. After 3 weeks, the animals were killed and the brains and spleens were removed. Rather unexpectedly, the tumors in several of the animals treated with CpG-ODN (both day 0 and day 3 group) were larger than in saline or m-CpG-ODN treated control animals. The tumor size in CpG-ODN-treated animals was more variable than in both control groups. This was associated with inflammatory responses and necrosis which was observed in most tumors following CpG treatment. This, however, did not prevent excessive growth of solid tumor masses in the CpG-treated animals similar to the control-treated animals. Dense infiltration with microglial cells resembling ramified microglia was observed within the solid tumor masses of control- and CpG-treated animals. In necrotic areas (phagocytic), activation of microglial cells was suggested by ED1 expression and a more macrophage-like morphology. Dense lymphocytic infiltrates consisting predominantly of CD8+ T cells and fewer NK cells were detected in all tumors including the control-treated animals. Expression of perforin serving as a marker for T cell or NK cell activation was detected only on isolated cells in all treatment groups. Tumors of all treatment groups revealed CD25 expression indicating T cells presumed to maintain peripheral tolerance to self-antigens. Cytotoxic T cell assays with in vitro restimulated lymphocytes ((51)chromium release assay) as well as interferon-gamma production by fresh splenocytes (Elispot assay) revealed specific responses to 9L cells but not another syngeneic cell line (MADB 106 adenocarcinoma). Surprisingly, the lysis rates with lymphocytes from CpG-ODN-treated animals were lower compared to control-treated animals. The tumor size of individual animals did not correlate with the response in both immune assays. Taken together, our data support the immunostimulatory capacity of CpG-ODN in normal brain. However, intratumoral application proved ineffective in a rat glioma model. CpG-ODN treatment may not yield beneficial effects in glioma patients.
Figures


Similar articles
-
Induction of anti-glioma natural killer cell response following multiple low-dose intracerebral CpG therapy.Clin Cancer Res. 2010 Jul 1;16(13):3399-408. doi: 10.1158/1078-0432.CCR-09-3087. Epub 2010 Jun 22. Clin Cancer Res. 2010. PMID: 20570924 Free PMC article.
-
Incomplete tumour control following DNA vaccination against rat gliomas expressing a model antigen.Acta Neurochir (Wien). 2013 Jan;155(1):51-8; discussion 59. doi: 10.1007/s00701-012-1526-7. Epub 2012 Nov 8. Acta Neurochir (Wien). 2013. PMID: 23132370 Free PMC article.
-
TLR ligands in the local treatment of established intracerebral murine gliomas.J Immunol. 2008 Nov 15;181(10):6720-9. doi: 10.4049/jimmunol.181.10.6720. J Immunol. 2008. PMID: 18981089
-
CpG oligodeoxynucleotide-based therapy of lymphoid malignancies.Adv Drug Deliv Rev. 2009 Mar 28;61(3):263-7. doi: 10.1016/j.addr.2008.12.006. Epub 2009 Jan 8. Adv Drug Deliv Rev. 2009. PMID: 19168102 Review.
-
Rat brain tumor models in experimental neuro-oncology: the C6, 9L, T9, RG2, F98, BT4C, RT-2 and CNS-1 gliomas.J Neurooncol. 2009 Sep;94(3):299-312. doi: 10.1007/s11060-009-9875-7. Epub 2009 Apr 21. J Neurooncol. 2009. PMID: 19381449 Free PMC article. Review.
Cited by
-
Human glioblastoma stem-like cells are more sensitive to allogeneic NK and T cell-mediated killing compared with serum-cultured glioblastoma cells.Brain Pathol. 2012 Mar;22(2):159-74. doi: 10.1111/j.1750-3639.2011.00515.x. Epub 2011 Sep 16. Brain Pathol. 2012. PMID: 21790828 Free PMC article.
-
Glioma-associated microglia/macrophages (GAMs) in glioblastoma: Immune function in the tumor microenvironment and implications for immunotherapy.Front Immunol. 2023 Mar 9;14:1123853. doi: 10.3389/fimmu.2023.1123853. eCollection 2023. Front Immunol. 2023. PMID: 36969167 Free PMC article. Review.
-
Gene Expression Profiling and Pathway Network Analysis Predicts a Novel Antitumor Function for a Botanical-Derived Drug, PG2.Evid Based Complement Alternat Med. 2015;2015:917345. doi: 10.1155/2015/917345. Epub 2015 Apr 20. Evid Based Complement Alternat Med. 2015. PMID: 25972907 Free PMC article.
-
CpG Oligodeoxynucleotides for Anticancer Monotherapy from Preclinical Stages to Clinical Trials.Pharmaceutics. 2021 Dec 28;14(1):73. doi: 10.3390/pharmaceutics14010073. Pharmaceutics. 2021. PMID: 35056969 Free PMC article. Review.
-
CpG-PEG Conjugates and their Immune Modulating Effects after Systemic Administration.Pharm Res. 2018 Mar 2;35(4):80. doi: 10.1007/s11095-018-2355-z. Pharm Res. 2018. PMID: 29500548
References
-
- Auf G, Carpentier AF, Chen L, Le Clanche C, Delattre JY. Implication of macrophages in tumor rejection induced by CpG-oligodeoxynucleotides without antigen. Clin Cancer Res. 2001;7:3540–3543. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

