Oxidative stress and glutathione in TGF-beta-mediated fibrogenesis
- PMID: 19800967
- PMCID: PMC2818240
- DOI: 10.1016/j.freeradbiomed.2009.09.026
Oxidative stress and glutathione in TGF-beta-mediated fibrogenesis
Abstract
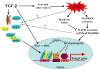
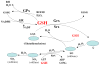
Transforming growth factor beta (TGF-beta) is the most potent and ubiquitous profibrogenic cytokine, and its expression is increased in almost all the fibrotic diseases and in experimental fibrosis models. TGF-beta increases reactive oxygen species production and decreases the concentration of glutathione (GSH), the most abundant intracellular free thiol and an important antioxidant, which mediates many of the fibrogenic effects of TGF-beta in various types of cells. A decreased GSH concentration is also observed in human fibrotic diseases and in experimental fibrosis models. Although the biological significance of GSH depletion in the development of fibrosis remains obscure, GSH and N-acetylcysteine, a precursor of GSH, have been used in clinics for the treatment of fibrotic diseases. This review summarizes recent findings in the field to address the potential mechanism whereby oxidative stress mediates fibrogenesis induced by TGF-beta and the potential therapeutic value of antioxidant treatment in fibrotic diseases.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures



References
-
- Dekhuijzen PN, Aben KK, Dekker I, Aarts LP, Wielders PL, van Herwaarden CL, Bast A. Increased exhalation of hydrogen peroxide in patients with stable and unstable chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1996;154:813–816. - PubMed
-
- Rahman I, Morrison D, Donaldson K, MacNee W. Systemic oxidative stress in asthma, COPD, and smokers. Am J Respir Crit Care Med. 1996;154:1055–1060. - PubMed
-
- Pratico D, Basili S, Vieri M, Cordova C, Violi F, Fitzgerald GA. Chronic obstructive pulmonary disease is associated with an increase in urinary levels of isoprostane F2alpha-III, an index of oxidant stress. Am J Respir Crit Care Med. 1998;158:1709–1714. - PubMed
-
- Barreiro E, de la Puente B, Minguella J, Corominas JM, Serrano S, Hussain SNA, Gea J. Oxidative Stress and Respiratory Muscle Dysfunction in Severe Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med. 2005;171:1116–1124. - PubMed
-
- Juul K, Tybjaerg-Hansen A, Marklund S, Lange P, Nordestgaard BG. Genetically Increased Antioxidative Protection and Decreased Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med. 2006;173:858–864. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

