Centralized modularity of N-linked glycosylation pathways in mammalian cells
- PMID: 19802388
- PMCID: PMC2750756
- DOI: 10.1371/journal.pone.0007317
Centralized modularity of N-linked glycosylation pathways in mammalian cells
Abstract
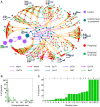
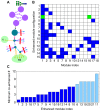
Glycosylation is a highly complex process to produce a diverse repertoire of cellular glycans that are attached to proteins and lipids. Glycans are involved in fundamental biological processes, including protein folding and clearance, cell proliferation and apoptosis, development, immune responses, and pathogenesis. One of the major types of glycans, N-linked glycans, is formed by sequential attachments of monosaccharides to proteins by a limited number of enzymes. Many of these enzymes can accept multiple N-linked glycans as substrates, thereby generating a large number of glycan intermediates and their intermingled pathways. Motivated by the quantitative methods developed in complex network research, we investigated the large-scale organization of such N-linked glycosylation pathways in mammalian cells. The N-linked glycosylation pathways are extremely modular, and are composed of cohesive topological modules that directly branch from a common upstream pathway of glycan synthesis. This unique structural property allows the glycan production between modules to be controlled by the upstream region. Although the enzymes act on multiple glycan substrates, indicating cross-talk between modules, the impact of the cross-talk on the module-specific enhancement of glycan synthesis may be confined within a moderate range by transcription-level control. The findings of the present study provide experimentally-testable predictions for glycosylation processes, and may be applicable to therapeutic glycoprotein engineering.
Conflict of interest statement
Figures
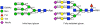
References
-
- Sinnott ML. Catalytic mechanism of enzymic glycosyl transfer. Chem Rev. 1990;90:1171–1202.
-
- Varki A, Cummings RD, Esko JD, Freeze HH, Hart GW, et al. New York: Cold Spring Harbor Laboratory Press; 2008. Essentials of glycobiology. - PubMed
-
- Raman R, Raguram S, Venkataraman G, Paulson JC, Sasisekharan R. Glycomics: an integrated systems approach to structure-function relationships of glycans. Nat Meth. 2005;2:817–824. - PubMed
-
- von der Lieth C-W, Bohne-Lang A, Lohmann KK, Frank M. Bioinformatics for glycomics: status, methods, requirements and perspectives. Brief Bioinform. 2004;5:164–178. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

