Feed-forward inhibition as a buffer of the neuronal input-output relation
- PMID: 19815518
- PMCID: PMC2764942
- DOI: 10.1073/pnas.0904784106
Feed-forward inhibition as a buffer of the neuronal input-output relation
Abstract
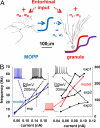
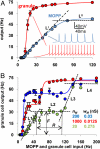
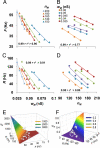
Neuronal processing depends on the input-output (I/O) relation between the frequency of synaptic stimulation and the resultant axonal firing rate. The all-or-none properties of spike generation and active membrane mechanisms can make the neuronal I/O relation very steep. The ensuing nearly bimodal behavior may severely limit information coding, as minimal input fluctuations within the expected natural variability could cause neuronal output to jump between quiescence and maximum firing rate. Here, using biophysically and anatomically realistic computational models of individual neurons, we demonstrate that feed-forward inhibition, a ubiquitous mechanism in which inhibitory interneurons and their target cells are activated by the same excitatory input, can change a steeply sigmoid I/O curve into a double-sigmoid typical of buffer systems. The addition of an intermediate plateau stabilizes the spiking response over a broad dynamic range of input frequency, ensuring robust integration of noisy synaptic signals. Both the buffered firing rate and its input firing range can be independently and extensively modulated by biologically plausible changes in the weight and number of excitatory synapses on the feed-forward interneuron. By providing a soft switch between essentially digital and analog rate-code, this continuous control of the circuit I/O could dramatically increase the computational power of neuronal integration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Persistent discharges in dentate gyrus perisoma-inhibiting interneurons require hyperpolarization-activated cyclic nucleotide-gated channel activation.J Neurosci. 2015 Mar 11;35(10):4131-9. doi: 10.1523/JNEUROSCI.3671-14.2015. J Neurosci. 2015. PMID: 25762660 Free PMC article.
-
Modulation of spike and burst rate in a minimal neuronal circuit with feed-forward inhibition.Neural Netw. 2013 Apr;40:1-17. doi: 10.1016/j.neunet.2012.12.008. Epub 2012 Dec 29. Neural Netw. 2013. PMID: 23376681
-
Rapid signaling at inhibitory synapses in a dentate gyrus interneuron network.J Neurosci. 2001 Apr 15;21(8):2687-98. doi: 10.1523/JNEUROSCI.21-08-02687.2001. J Neurosci. 2001. PMID: 11306622 Free PMC article.
-
Interneuron networks in the hippocampus.Curr Opin Neurobiol. 2011 Oct;21(5):709-16. doi: 10.1016/j.conb.2011.05.006. Epub 2011 Jun 1. Curr Opin Neurobiol. 2011. PMID: 21636266 Review.
-
Role of microcircuit structure and input integration in hippocampal interneuron recruitment and plasticity.Neuropharmacology. 2011 Apr;60(5):730-9. doi: 10.1016/j.neuropharm.2010.12.017. Epub 2010 Dec 30. Neuropharmacology. 2011. PMID: 21195097 Review.
Cited by
-
Association with reward negatively modulates short latency phasic conditioned responses of dorsal raphe nucleus neurons in freely moving rats.J Neurosci. 2013 Mar 13;33(11):5065-78. doi: 10.1523/JNEUROSCI.5679-12.2013. J Neurosci. 2013. PMID: 23486976 Free PMC article.
-
Modulation of medial prefrontal cortical activity using in vivo recordings and optogenetics.Mol Brain. 2012 Oct 8;5:36. doi: 10.1186/1756-6606-5-36. Mol Brain. 2012. PMID: 23044043 Free PMC article.
-
Signal denoising through topographic modularity of neural circuits.Elife. 2023 Jan 26;12:e77009. doi: 10.7554/eLife.77009. Elife. 2023. PMID: 36700545 Free PMC article.
-
Metabotropic Glutamate Receptor 7: From Synaptic Function to Therapeutic Implications.Curr Neuropharmacol. 2016;14(5):504-13. doi: 10.2174/1570159x13666150716165323. Curr Neuropharmacol. 2016. PMID: 27306064 Free PMC article. Review.
-
Cognitive control persistently enhances hippocampal information processing.Nature. 2021 Dec;600(7889):484-488. doi: 10.1038/s41586-021-04070-5. Epub 2021 Nov 10. Nature. 2021. PMID: 34759316 Free PMC article.
References
-
- Jonas P, et al. Interneuron diversity series: Fast in, fast out–temporal and spatial signal processing in hippocampal interneurons. Trends Neurosci. 2004;27:30–40. - PubMed
-
- Holt GR, Koch C. Shunting inhibition does not have a divisive effect on firing rates. Neural Comput. 1997;9:1001–1013. - PubMed
-
- Mitchell SJ, Silver RA. Shunting inhibition modulates neuronal gain during synaptic excitation. Neuron. 2003;38:433–445. - PubMed
-
- Gupta A, Wang Y, Markram H. Organizing principles for a diversity of GABAergic interneurons and synapses in the neocortex. Science. 2000;287:273–278. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

