TOC1 functions as a molecular switch connecting the circadian clock with plant responses to drought
- PMID: 19816401
- PMCID: PMC2790485
- DOI: 10.1038/emboj.2009.297
TOC1 functions as a molecular switch connecting the circadian clock with plant responses to drought
Abstract
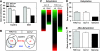
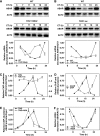
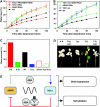
Despite our increasing knowledge on the transcriptional networks connecting abscisic acid (ABA) signalling with the circadian clock, the molecular nodes in which both pathways converge to translate the environmental information into a physiological response are not known. Here, we provide evidence of a feedback mechanism linking the circadian clock with plant responses to drought. A key clock component (TOC1, timing of CAB expression 1) binds to the promoter of the ABA-related gene (ABAR/CHLH/GUN5) and controls its circadian expression. TOC1 is in turn acutely induced by ABA and this induction advances the phase of TOC1 binding and modulates ABAR circadian expression. Moreover, the gated induction of TOC1 by ABA is abolished in ABAR RNAi plants suggesting that the reciprocal regulation between ABAR and TOC1 expression is important for sensitized ABA activity. Genetic studies with TOC1 and ABAR over-expressing and RNAi plants showed defective responses to drought, which support the notion that clock-dependent gating of ABA function is important for cellular homeostasis under dry environments.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Comment in
-
A functional connection between the clock component TOC1 and abscisic acid signaling pathways.Plant Signal Behav. 2010 Apr;5(4):409-11. doi: 10.4161/psb.5.4.11213. Epub 2010 Apr 14. Plant Signal Behav. 2010. PMID: 20339316 Free PMC article.
References
-
- Alabadí D, Oyama T, Yanovsky MJ, Harmon FG, Más P, Kay SA (2001) Reciprocal regulation between TOC1 and LHY/CCA1 within the Arabidopsis circadian clock. Science 293: 880–883 - PubMed
-
- Busk PK, Pagès M (1998) Regulation of abscisic acid-induced transcription. Plant Mol Biol 37: 425–435 - PubMed
-
- Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16: 735–743 - PubMed
-
- Correia MJ, Pereira JS, Chaves MM, Rodrigues ML, Pacheco CA (1995) ABA xylem concentrations determine maximum daily leaf conductance of field-grown Vitis vinifera L. plants. Plant Cell Environ 18: 511–521
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

