Epigenetic reversion of post-implantation epiblast to pluripotent embryonic stem cells
- PMID: 19816418
- PMCID: PMC3863718
- DOI: 10.1038/nature08534
Epigenetic reversion of post-implantation epiblast to pluripotent embryonic stem cells
Abstract
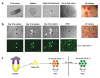
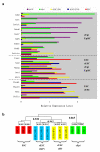
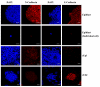
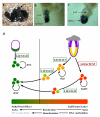
The pluripotent state, which is first established in the primitive ectoderm cells of blastocysts, is lost progressively and irreversibly during subsequent development. For example, development of post-implantation epiblast cells from primitive ectoderm involves significant transcriptional and epigenetic changes, including DNA methylation and X chromosome inactivation, which create a robust epigenetic barrier and prevent their reversion to a primitive-ectoderm-like state. Epiblast cells are refractory to leukaemia inhibitory factor (LIF)-STAT3 signalling, but they respond to activin/basic fibroblast growth factor to form self-renewing epiblast stem cells (EpiSCs), which exhibit essential properties of epiblast cells and that differ from embryonic stem (ES) cells derived from primitive ectoderm. Here we show reprogramming of advanced epiblast cells from embryonic day 5.5-7.5 mouse embryos with uniform expression of N-cadherin and inactive X chromosome to ES-cell-like cells (rESCs) in response to LIF-STAT3 signalling. Cultured epiblast cells overcome the epigenetic barrier progressively as they proceed with the erasure of key properties of epiblast cells, resulting in DNA demethylation, X reactivation and expression of E-cadherin. The accompanying changes in the transcriptome result in a loss of phenotypic and epigenetic memory of epiblast cells. Using this approach, we report reversion of established EpiSCs to rESCs. Moreover, unlike epiblast and EpiSCs, rESCs contribute to somatic tissues and germ cells in chimaeras. Further studies may reveal how signalling-induced epigenetic reprogramming may promote reacquisition of pluripotency.
Figures

References
-
- Gardner RL, Rossant J. Investigation of the fate of 4-5 day post-coitum mouse inner cell mass cells by blastocyst injection. J. Embryol. Exp. Morphol. 1979;52:141–152. - PubMed
-
- Surani MA, Hayashi K, Hajkova P. Genetic and epigenetic regulators of pluripotency. Cell. 2007;128:747–762. - PubMed
-
- Tesar PJ, et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. 2007;448:196–199. - PubMed
-
- Brons IG, et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature. 2007;448:191–195. - PubMed
-
- Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

