Dispersible formulation of artemether/lumefantrine: specifically developed for infants and young children
- PMID: 19818174
- PMCID: PMC2760242
- DOI: 10.1186/1475-2875-8-S1-S7
Dispersible formulation of artemether/lumefantrine: specifically developed for infants and young children
Abstract
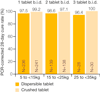
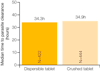
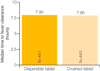
Infants and children under five years of age are the most vulnerable to malaria with over 1,700 deaths per day from malaria in this group. However, until recently, there were no WHO-endorsed paediatric anti-malarial formulations available. Artemisinin-based combination therapy is the current standard of care for patients with uncomplicated falciparum malaria in Africa. Artemether/lumefantrine (AL) meets WHO pre-qualification criteria for efficacy, safety and quality. Coartem, a fixed dose combination of artemether and lumefantrine, has consistently achieved cure rates of >95% in clinical trials. However, AL tablets are inconvenient for caregivers to administer as they need to be crushed and mixed with water or food for infants and young children. Further, in common with other anti-malarials, they have a bitter taste, which may result in children spitting the medicine out and not receiving the full therapeutic dose. There was a clear unmet medical need for a formulation of AL specifically designed for children. Ahead of a call from WHO for child-friendly medicines, Novartis, working in partnership with Medicines for Malaria Venture (MMV), started the development of a new formulation of AL for infants and young children: Coartem Dispersible. The excellent efficacy, safety and tolerability already demonstrated by AL tablets were confirmed with dispersible AL in a large trial comparing the crushed tablets with dispersible tablets in 899 African children with falciparum malaria. In the evaluable population, 28-day PCR-corrected cure rates of >96% were achieved. Further, its sweet taste means that it is palatable for children, and the dispersible formulation makes it easier for caregivers to administer than bitter crushed tablets. Easing administration may foster compliance, hence improving therapeutic outcomes in infants and young children and helping to preserve the efficacy of ACT.
Figures


References
-
- World Health Organization Make medicines child size http://www.who.int/childmedicines/en/
-
- World Health Organization 10 facts on children and medicines http://www.who.int/childmedicines/facts/en/index.html
-
- Juma EA, Obonyo CO, Akhwale WS, Ogutu BR. A randomized, open-label, comparative efficacy trial of artemether-lumefantrine suspension versus artemether-lumefantrine tablets for treatment of uncomplicated Plasmodium falciparum malaria in children in western Kenya. Malar J. 2008;7:262. doi: 10.1186/1475-2875-7-262. - DOI - PMC - PubMed
-
- World Health Organization Guidelines for the treatment of malaria http://apps.who.int/malaria/treatmentguidelines.html
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

