Consciousness and epilepsy: why are complex-partial seizures complex?
- PMID: 19818900
- PMCID: PMC2901990
- DOI: 10.1016/S0079-6123(09)17711-7
Consciousness and epilepsy: why are complex-partial seizures complex?
Abstract
Why do complex-partial seizures in temporal lobe epilepsy (TLE) cause a loss of consciousness? Abnormal function of the medial temporal lobe is expected to cause memory loss, but it is unclear why profoundly impaired consciousness is so common in temporal lobe seizures. Recent exciting advances in behavioral, electrophysiological, and neuroimaging techniques spanning both human patients and animal models may allow new insights into this old question. While behavioral automatisms are often associated with diminished consciousness during temporal lobe seizures, impaired consciousness without ictal motor activity has also been described. Some have argued that electrographic lateralization of seizure activity to the left temporal lobe is most likely to cause impaired consciousness, but the evidence remains equivocal. Other data correlates ictal consciousness in TLE with bilateral temporal lobe involvement of seizure spiking. Nevertheless, it remains unclear why bilateral temporal seizures should impair responsiveness. Recent evidence has shown that impaired consciousness during temporal lobe seizures is correlated with large-amplitude slow EEG activity and neuroimaging signal decreases in the frontal and parietal association cortices. This abnormal decreased function in the neocortex contrasts with fast polyspike activity and elevated cerebral blood flow in limbic and other subcortical structures ictally. Our laboratory has thus proposed the "network inhibition hypothesis," in which seizure activity propagates to subcortical regions necessary for cortical activation, allowing the cortex to descend into an inhibited state of unconsciousness during complex-partial temporal lobe seizures. Supporting this hypothesis, recent rat studies during partial limbic seizures have shown that behavioral arrest is associated with frontal cortical slow waves, decreased neuronal firing, and hypometabolism. Animal studies further demonstrate that cortical deactivation and behavioral changes depend on seizure spread to subcortical structures including the lateral septum. Understanding the contributions of network inhibition to impaired consciousness in TLE is an important goal, as recurrent limbic seizures often result in cortical dysfunction during and between epileptic events that adversely affects patients' quality of life.
Figures
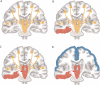
 ). (B) Simple-partial seizures arising from the temporal lobe are not associated with widespread CBF changes (n =
). (B) Simple-partial seizures arising from the temporal lobe are not associated with widespread CBF changes (n =  ). For (A) and (B), extent threshold, k =
). For (A) and (B), extent threshold, k =  voxels (voxel size =
voxels (voxel size =  × 2 × 2 mm). Height threshold, P =
× 2 × 2 mm). Height threshold, P =  . Equivalently, only voxel clusters greater than 1 cm3 in volume and with Z scores greater than 2.33 are displayed. Adapted with permission from Blumenfeld et al. (2004a). Please see online version of this article for full color figure.
. Equivalently, only voxel clusters greater than 1 cm3 in volume and with Z scores greater than 2.33 are displayed. Adapted with permission from Blumenfeld et al. (2004a). Please see online version of this article for full color figure.
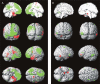
 Cg1, anterior cingulate cortex; HC, hippocampus; OFC, orbitofrontal cortex; RSC, retrosplenial/posterior cingulate cortex; Thal, thalamus. Adapted with permission from Englot et al. (2008). Please see online version of this article for full color figure.
Cg1, anterior cingulate cortex; HC, hippocampus; OFC, orbitofrontal cortex; RSC, retrosplenial/posterior cingulate cortex; Thal, thalamus. Adapted with permission from Englot et al. (2008). Please see online version of this article for full color figure.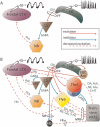
Similar articles
-
Impaired consciousness in temporal lobe seizures: role of cortical slow activity.Brain. 2010 Dec;133(Pt 12):3764-77. doi: 10.1093/brain/awq316. Epub 2010 Nov 16. Brain. 2010. PMID: 21081551 Free PMC article.
-
Focal Impaired Awareness Seizure.2024 Mar 19. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 19. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30085572 Free Books & Documents.
-
Spectral and spatial shifts of post-ictal slow waves in temporal lobe seizures.Brain. 2012 Oct;135(Pt 10):3134-43. doi: 10.1093/brain/aws221. Epub 2012 Aug 24. Brain. 2012. PMID: 22923634 Free PMC article.
-
Theories of impaired consciousness in epilepsy.Ann N Y Acad Sci. 2009 Mar;1157:48-60. doi: 10.1111/j.1749-6632.2009.04472.x. Ann N Y Acad Sci. 2009. PMID: 19351355 Free PMC article. Review.
-
Ictal vomiting as a sign of temporal lobe epilepsy confirmed by stereo-EEG and surgical outcome.Epilepsy Behav. 2015 Dec;53:112-6. doi: 10.1016/j.yebeh.2015.10.009. Epub 2015 Nov 8. Epilepsy Behav. 2015. PMID: 26558713 Review.
Cited by
-
Abnormal functional connectivity of the posterior hypothalamus and other arousal regions in surgical temporal lobe epilepsy.J Neurosurg. 2023 Feb 17;139(3):640-650. doi: 10.3171/2023.1.JNS221452. Print 2023 Sep 1. J Neurosurg. 2023. PMID: 36807210 Free PMC article.
-
Consciousness as a useful concept in epilepsy classification.Epilepsia. 2014 Aug;55(8):1145-50. doi: 10.1111/epi.12588. Epub 2014 Jun 30. Epilepsia. 2014. PMID: 24981294 Free PMC article. Review.
-
Transient Epileptic Amnesia with Preserved Consciousness: a Report of Two Cases.J Epilepsy Res. 2017 Jun 30;7(1):54-56. doi: 10.14581/jer.17010. eCollection 2017 Jun. J Epilepsy Res. 2017. PMID: 28775957 Free PMC article.
-
Role of the Nucleus Basalis as a Key Network Node in Temporal Lobe Epilepsy.Neurology. 2021 Mar 2;96(9):e1334-e1346. doi: 10.1212/WNL.0000000000011523. Epub 2021 Jan 13. Neurology. 2021. PMID: 33441453 Free PMC article.
-
A prospective study of loss of consciousness in epilepsy using virtual reality driving simulation and other video games.Epilepsy Behav. 2010 Jul;18(3):238-46. doi: 10.1016/j.yebeh.2010.04.011. Epilepsy Behav. 2010. PMID: 20537593 Free PMC article.
References
-
- Alarcon G, Elwes RDC, Polkey CE, Binnie CD. Ictal oroalimentary automatisms with preserved consciousness: Implications for the pathophysiology of automatisms and relevance to the international classification of seizures. Epilepsia. 1998;39:1119–1122. - PubMed
-
- Albert ML, Silverberg R, Reches A, Berman M. Cerebral dominance for consciousness. Archives of Neurology. 1976;33:453–454. - PubMed
-
- Andersen AR, Waldemar G, Dam M, Fuglsang-Frederiksen A, Herning M, Kruse-Larsen C. SPECT in the presurgical evaluation of patients with temporal lobe epilepsy — A preliminary report. Acta Neurochirurgica Supplementum. 1990;50:80–83. - PubMed
-
- Angelucci A, Levitt JB, Lund JS. Anatomical origins of the classical receptive field and modulatory surround field of single neurons in macaque visual cortical area V1. Progress in Brain Research. 2002;136:373–388. - PubMed
-
- Ardila A. Partial cognitive seizures. Neuropsychiatry, Neuropsychology, and Behavioral Neurology. 1990;2:175–182.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

