Novel mechanisms of protection against acetaminophen hepatotoxicity in mice by glutathione and N-acetylcysteine
- PMID: 19821517
- PMCID: PMC2977522
- DOI: 10.1002/hep.23267
Novel mechanisms of protection against acetaminophen hepatotoxicity in mice by glutathione and N-acetylcysteine
Abstract
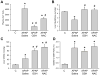
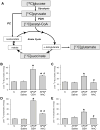
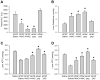
Acetaminophen (APAP) overdose is a major cause of acute liver failure. The glutathione (GSH) precursor N-acetylcysteine (NAC) is used to treat patients with APAP overdose for up to 48 hours. Although it is well established that early treatment with NAC can improve the scavenging of the reactive metabolite N-acetyl-p-benzoquinone imine, protective mechanisms at later times remain unclear. To address this issue, fasted C3Heb/FeJ mice were treated with 300 mg/kg APAP and then received intravenously 0.65 mmol/kg GSH or NAC at 1.5 hours after APAP. The animals were sacrificed at 6 hours. APAP alone caused severe liver injury with peroxynitrite formation and DNA fragmentation, all of which was attenuated by both treatments. However, GSH (-82%) was more effective than NAC (-46%) in preventing liver injury. Using nuclear magnetic resonance spectroscopy to measure tissue adenosine triphosphate (ATP) levels and the substrate flux through the mitochondrial Krebs cycle, it was observed that the reduced liver injury correlated with accelerated recovery of mitochondrial GSH content, maintenance of ATP levels, and an increased substrate supply for the mitochondrial Krebs cycle compared with APAP alone. NAC treatment was less effective in recovering ATP and mitochondrial GSH levels and showed reduced substrate flux through the Krebs cycle compared with GSH. However, increasing the dose of NAC improved the protective effect similar to GSH, suggesting that the amino acids not used for GSH synthesis were used as mitochondrial energy substrates.
Conclusion: Delayed treatment with GSH and NAC protect against APAP overdose by dual mechanisms-that is, by enhancing hepatic and mitochondrial GSH levels (scavenging of reactive oxygen and peroxynitrite)-and by supporting the mitochondrial energy metabolism.
Figures



References
-
- Larson AM, Polson J, Fontana RJ, Davern TJ, Lalani E, Hynan LS, et al. Acute Liver Failure Study Group. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. HEPATOLOGY. 2005;42:1364–1372. - PubMed
-
- Mitchell JR, Jollow DJ, Potter WZ, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. J Pharmacol Exp Ther. 1973;187:211–217. - PubMed
-
- Jollow DJ, Mitchell JR, Potter WZ, Davis DC, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis. II. Role of covalent binding in vivo. J Pharmacol Exp Ther. 1973;187:195–202. - PubMed
-
- Mitchell JR, Jollow DJ, Potter WZ, Davis DC, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis. I. Role of drug metabolism. J Pharmacol Exp Ther. 1973;187:185–194. - PubMed
-
- Prescott LF, Park J, Ballantyne A, Adriaenssens P, Proudfoot AT. Treatment of paracetamol (acetaminophen) poisoning with N-acetylcysteine. Lancet. 1977;2:432–434. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
